Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
#1876 2026-02-22 18:00:01
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2439) Cyril N. Hinshelwood
Gist:
Work
During chemical reactions, atoms and molecules regroup and form new constellations. When molecules formed during a reaction readily react with molecules present from the beginning, a chain reaction can occur. Explosions and fire are examples of chain reactions. During the 1930s Cyril Hinshelwood analyzed conditions and sequences of events involved in chain reactions from a theoretical standpoint. Among other things, he found that the theoretical results corresponded with observations of the reaction between hydrogen and oxygen.
Summary
Sir Cyril Norman Hinshelwood (born June 19, 1897, London, Eng.—died Oct. 9, 1967, London) was a British chemist who worked on reaction rates and reaction mechanisms, particularly that of the combination of hydrogen and oxygen to form water, one of the most fundamental combining reactions in chemistry. For this work he shared the 1956 Nobel Prize for Chemistry with the Soviet scientist Nikolay Semyonov.
Hinshelwood obtained his doctorate at the University of Oxford in 1924 and became professor of chemistry there in 1937. After retiring from Oxford in 1964 he became a senior research fellow at Imperial College, London.
About 1930 Hinshelwood began investigating the complex reaction in which hydrogen and oxygen atoms combine to form water. He showed that the products of this reaction help to spread the reaction further in what is essentially a chain reaction.
He next sought to explore molecular kinetics within the bacterial cell. Upon observing the biological responses of bacteria to changes in environment, he concluded that more or less permanent changes in a cell’s resistance to a drug could be induced. This finding was important in regard to bacterial resistance to antibiotic and other chemotherapeutic agents. Hinshelwood was knighted in 1948. His publications include The Kinetics of Chemical Change in Gaseous Systems (1926) and The Chemical Kinetics of the Bacterial Cell (1946).
Details
Sir Cyril Norman Hinshelwood (19 June 1897 – 9 October 1967) was a British physical chemist and expert in chemical kinetics. His work in reaction mechanisms earned the 1956 Nobel Prize in chemistry.
Education
Born in London, his parents were Norman Macmillan Hinshelwood, a chartered accountant, and Ethel Frances née Smith. He was educated first in Canada, returning in 1905 on the death of his father to a small flat in Chelsea where he lived for the rest of his life. He then studied at Westminster City School and Balliol College, Oxford.
Career
During the First World War, Hinshelwood was a chemist in an explosives factory. He was a tutor at Trinity College, Oxford, from 1921 to 1937 and was Dr Lee's Professor of Chemistry at the University of Oxford from 1937. He served on several advisory councils on scientific matters to the British Government.
His early studies of molecular kinetics led to the publication of Thermodynamics for Students of Chemistry and The Kinetics of Chemical Change in 1926. With Harold Warris Thompson he studied the explosive reaction of hydrogen and oxygen and described the phenomenon of chain reaction. His subsequent work on chemical changes in the bacterial cell proved to be of great importance in later research work on antibiotics and therapeutic agents, and his book, The Chemical Kinetics of the Bacterial Cell was published in 1946, followed by Growth, Function and Regulation in Bacterial Cells in 1966. In 1951 he published The Structure of Physical Chemistry. It was republished as an Oxford Classic Texts in the Physical Sciences by Oxford University Press in 2005.
The Langmuir-Hinshelwood process in heterogeneous catalysis, in which the adsorption of the reactants on the surface is the rate-limiting step, is named after him. He was a senior research fellow at Imperial College London from 1964 to 1967.
Awards and honours
In addition to being named the second Dr. Lee's Professor of Chemistry at Oxford, Hinshelwood was elected Fellow of the Royal Society (FRS) in 1929, serving as president from 1955 to 1960. He was knighted in 1948 and appointed to the Order of Merit in 1960. With Nikolay Semenov of the USSR, Hinshelwood was jointly awarded the Nobel Prize in Chemistry in 1956 for his researches into the mechanism of chemical reactions. He was also an elected member of the American Academy of Arts and Sciences, the United States National Academy of Sciences, and the American Philosophical Society.
Hinshelwood was president of the Chemical Society, the Royal Society, the Classical Association, and the Faraday Society, and received numerous awards and honorary degrees. He was elected on 1 January 1960 to honorary membership of the Manchester Literary and Philosophical Society who awarded him its Dalton medal in 1966.
Personal life
Hinshelwood never married. He was fluent in seven classical and modern languages and his main hobbies were painting, collecting Chinese pottery, and foreign literature. As an artist, Hinshelwood painted scenes in Oxford, as well as portraits of Oxford University people including Harold Hartley, his doctoral supervisor, and Herbert Blakiston, the President of Trinity College. The portrait of Hartley is now owned by the Royal Society, and that of Blakiston is owned by Trinity College, as are a number of Hinshelwood's other paintings.
He died, at home, on 9 October 1967. In 1968, his Nobel Prize medal was sold by his estate to a collector, who then sold it in 1976 for $15,000. In 2017, his Nobel Prize medal was sold at auction for $128,000.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1877 2026-02-23 17:37:59
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2440) Nikolay Semyonov
Gist:
Work
During chemical reactions, atoms and molecules regroup and form new constellations. When molecules formed during a reaction readily react with molecules present from the beginning, a chain reaction can occur. Explosions and fire are examples of chain reactions. During the 1930s Nikolay Semyonov analyzed conditions and sequences of events involved in chain reactions from a theoretical and mathematical standpoint. Among other things, he found that the theoretical results corresponded with observations of the reactions between phosphorus and oxygen and between hydrogen and oxygen.
Work
During chemical reactions, atoms and molecules regroup and form new constellations. When molecules formed during a reaction readily react with molecules present from the beginning, a chain reaction can occur. Explosions and fire are examples of chain reactions. During the 1930s Nikolay Semyonov analyzed conditions and sequences of events involved in chain reactions from a theoretical and mathematical standpoint. Among other things, he found that the theoretical results corresponded with observations of the reactions between phosphorus and oxygen and between hydrogen and oxygen.
Summary
Nikolay Nikolayevich Semyonov (born April 15 [April 3, Old Style], 1896, Saratov, Russia—died Sept. 25, 1986, Moscow, U.S.S.R.) was a Soviet physical chemist who shared the 1956 Nobel Prize for Chemistry with Sir Cyril Hinshelwood for research in chemical kinetics. He was the second Soviet citizen (after the émigré writer Ivan Bunin) to receive a Nobel Prize.
Semyonov was educated in St. Petersburg, graduating from the city’s university in 1917, the year of the Russian Revolution, and taught for a time at the University of Tomsk in western Siberia. Associated with the Leningrad A.F. Ioffe Physicotechnical Institute from 1920 to 1931, he became a professor at the Leningrad (St. Petersburg) Polytechnic Institute in 1928. He was director of the Institute of Chemical Physics at the Academy of Sciences of the U.S.S.R. after 1931 and became a professor at Moscow State University in 1944.
Like Hinshelwood, Semyonov conducted research on the mechanism of chemical chain reactions and their significance in relation to explosions. Semyonov was the first to show that chain reactions are the norm in chemical transformations of matter. He published the influential book O nekotorykh problemakh khimicheskoy kinetiki i reaktsionnoy sposobnosti (1954; Some Problems in Chemical Kinetics and Reactivity).
Details
Nikolay Nikolayevich Semyono , sometimes Semenov, Semionov or Semenoff (15 April [O.S. 3 April] 1896 – 25 September 1986) was a Soviet physicist and chemist. Semyonov was awarded the 1956 Nobel Prize in Chemistry for his work on the mechanism of chemical transformation.
Life and career
Semyonov was born in Saratov, the son of Yelena Dmitrieva and Nikolai Aleksandrovich Semyonov. He graduated from the department of physics of Petrograd University (1913–1917), where he was a student of Abram Fyodorovich Ioffe. In 1918, he moved to Samara, where he was enlisted into Kolchak's White Army during Russian Civil War.
Semyonov published his first research paper in 1916 and became a lecturer at the University of Tomsk in western Siberia.
After graduating from Saint Petersburg State University, he worked as an assistant and lecturer at the Tomsk and Tomsk University Institute of Technology, where he published his first research paper in 1916. He returned to western Siberia, Petrograd and took charge of the electron phenomena laboratory of the Petrograd Physico-Technical Institute in 1920. He also became the vice-director of the institute. In 1921, he married philologist Maria Boreishe-Liverovsky (student of Zhirmunsky). She died two years later. On September 15, 1924, Nikolay married Maria's niece, Natalia Nikolayevna Burtseva. They had two children, son Yuri and daughter Lyudmila.
During that difficult time, Semyonov, together with Pyotr Kapitsa, discovered a way to measure the magnetic field of an atomic nucleus (1922). Later the experimental setup was improved by Otto Stern and Walther Gerlach and became known as Stern–Gerlach experiment.
In 1925, Semyonov, together with Yakov Frenkel, studied kinetics of condensation and adsorption of vapors. In 1927, he studied ionisation in gases and published an important book, Chemistry of the Electron. In 1928, he, together with Vladimir Fock, created a theory of thermal disruptive discharge of dielectrics.
In 1927, Semyonov studied the ionization of gases, the chemistry of the electron. In 1928, he created the theory of the broken discharge of dielectrics with Vladimir Fock.
He lectured at the Petrograd Polytechnical Institute and was appointed Professor in 1928. In 1931, he organized the Institute of Physical Chemistry of the USSR Academy of Sciences (which moved to Chernogolovka in 1943) and became its first director. In 1932, he became a full member of the Soviet Academy of Sciences.
The ideas of Semyonov have been applied in the science of reaction and production of polymerization reactions. His ideas are also applied in catalysis studies in biological systems.
Semyonov married Natalya Nikolayevna Semyonov and together they both have a son and a daughter. Semyonov died on September 25, 1986, in Moscow, and was buried at the Novodevichy Cemetery.
Significant works
Semyonov's outstanding work on the mechanism of chemical transformation includes an exhaustive analysis of the application of the chain theory to varied reactions (1934–1954) and, more significantly, to combustion processes. He proposed a theory of degenerate branching, which led to a better understanding of the phenomena associated with the induction periods of oxidation processes. He spent most of his career focusing and developing the field of chemical chain reactions.
Semyonov wrote two important books outlining his work. Chemical Kinetics and Chain Reactions was published in 1934, with an English edition in 1935. It was the first book in the U.S.S.R. to develop a detailed theory of unbranched and branched chain reactions in chemistry. Some Problems of Chemical Kinetics and Reactivity, first published in 1954, was revised in 1958; there are also English, American, German, and Chinese editions. He is the only Soviet/Russian Chemistry Nobel Laureate, who received the Nobel Prize in Chemistry (together with Sir Cyril N. Hinshelwood) for his work in 1956.
Semyonov had long been a supporter of the Communist Party and the Soviet Union. After the Bulletin of the Atomic Scientists accused the Soviet Union of heavy scientific censorship in 1953, he coauthored the Soviet response which denied all accusations. He is also noted as being the most famous signatory to a 1971 public letter from Soviet scientists to United States president Richard Nixon, on displeasure in the murder trial of Angela Davis.
Semyonov trained Russian organometallic chemist Alexander Shilov, who discovered platinum catalyzed C-H activation.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1878 2026-02-25 00:16:38
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2441) André Frédéric Cournand
Gist:
Life
André Cournand was born in Paris. His father was a doctor, and Cournand studied natural science and medicine in Paris. After becoming a medical doctor, he made his way to the U.S. in 1930 to spend a year at Bellevue Hospital and Columbia University in New York. There he began a prolonged collaboration with Dickinson Richards and decided to stay in the U.S. Cournand married Sibylle Blumer, who was a widow, and adopted her son. The couple had three more children.
Work
Even though Werner Forssmann succeeded in inserting a catheter into his own heart in 1929, there was great hesitance about continuing this type of research. Nonetheless, beginning in 1941 André Cournand and Dickinson Richards published a series of studies that established use of cardiac catheterization, among other things, to introduce contrast fluid for X-ray images and to measure pressure and oxygen content. Because it was possible to reach the upper chambers of the heart, blood pressure and the blood’s oxygen content could be measured on the way from the heart to the lungs, which was impossible before.
Summary
André F. Cournand (born Sept. 24, 1895, Paris, France—died Feb. 19, 1988, Great Barrington, Mass., U.S.) was a French-American physician and physiologist who in 1956 shared the Nobel Prize for Physiology or Medicine with Dickinson W. Richards and Werner Forssmann for discoveries concerning heart catheterization and circulatory changes.
His medical studies interrupted by World War I, Cournand graduated from the University of Paris in 1930. He studied at Bellevue Hospital, New York City, where he met Richards. Together they collaborated in clinical lung and heart research and perfected Forssmann’s procedure, now termed cardiac catheterization, whereby a tube is passed into the heart from a vein at the elbow. With this procedure it became possible to study the functioning of the diseased human heart and to make more accurate diagnoses of the underlying anatomic defects. Cournand and Richards also used the catheter to examine the pulmonary artery, thus enabling improvement in the diagnosis of lung diseases as well.
Cournand joined the faculty of the College of Physicians and Surgeons of Columbia University in 1934, retiring as emeritus professor of medicine in 1964. He remained active as a special lecturer until his final illness. He became a naturalized citizen of the United States in 1941.
Details
André Frédéric Cournand (September 24, 1895 – February 19, 1988) was a French-American physician and physiologist.
Biography
Cournand was awarded the Nobel Prize in Physiology or Medicine in 1956 along with Werner Forssmann and Dickinson W. Richards for the development of cardiac catheterization.
Born in Paris, Cournand emigrated to the United States in 1930 and, in 1941, became a naturalized citizen. For most of his career, Cournand was a professor at the Columbia University College of Physicians and Surgeons and worked at Bellevue Hospital in New York City.
Many seats of medical research have recognized his work, and he has received the Anders Retzius Silver Medal of the Swedish Society for Internal Medicine (1946), the Albert Lasker Award for Basic Medical Research (1949), the John Philipps Memorial Award of the American College of Physicians (1952), the Gold Medal of the Académie Royale de Médecine de Belgique and of the Académie Nationale de Médecine, Paris (1956). He was elected Doctor (honoris causa) of the Universities of Strasbourg (1957), Lyon (1958), Brussels (1959), Pisa (1961), and D.Sc. of the University of Birmingham (1961).
In 1981, Cournand became a founding member of the World Cultural Council.
His widow Beatrice died in 1993 aged 90.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1879 2026-02-26 00:21:04
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2442) Werner Forssmann
Gist:
Life
Werner Forssmann was born in Berlin, where he also studied medicine. As a newly educated doctor, he served in Eberswalde and conducted his Nobel Prize-awarded experiment there in 1929. His experimentation met resistance, however, which impeded continued research in the field. After being chief surgeon in Dresden and Berlin, Forssmann served as a doctor in the army during World War II. After the war ended, he worked as a district medical officer, among other things. Forssmann and his wife, also a doctor, had six children.
Work
In 1929 the physician Werner Forssmann saw a picture in a book showing how a tube was inserted into the heart of a horse through a vein. A balloon at the other end of the tube showed changes in pressure. Forssmann was convinced that a similar experiment could be carried out on people. Despite the fact that his boss forbade him, Forssmann conducted the experiment on himself. From the crook of his arm he inserted a thin catheter through a vein into his heart and took an X-ray photo. The experiment paved the way for many types of heart studies.
Summary
Werner Forssmann (born Aug. 20, 1904, Berlin, Ger.—died June 1, 1979, Schopfheim, W. Ger.) was a German surgeon who shared with André F. Cournand and Dickinson W. Richards the Nobel Prize for Physiology or Medicine in 1956. A pioneer in heart research, Forssmann contributed to the development of cardiac catheterization, a procedure in which a tube is inserted into a vein at the elbow and passed through the vein into the heart. While a surgical resident in Berlin (1929), Forssmann used himself as the first human subject, watching the progress of the catheter in a mirror held in front of a fluoroscope screen. Forssmann’s daring experiment was condemned at the time as foolhardy and dangerous, and in the face of severe criticism he abandoned cardiology for urology.
Forssmann’s procedure, with slight modifications, was put into practice in 1941 by Richards and Cournand, and has since become an extremely valuable tool in diagnosis and research. It has made possible, among other things, precise measurement of intracardiac pressure and blood flow, injection into the heart of drugs and of opaque material visible on X-ray photographs, and insertion of electrodes for the regulation of the heartbeat.
Forssmann graduated in medicine from the University of Berlin (1928) and then did postgraduate study in urology at Berlin and Mainz. He served as chief of surgery at the city hospital in Dresden-Friedrichstadt and in 1958 was named chief of the surgical division of the Evangelical Hospital in Düsseldorf.
Details
Werner Theodor Otto Forßmann (29 August 1904 – 1 June 1979) was a German researcher and physician from Germany who shared the 1956 Nobel Prize in Medicine (with Andre Frederic Cournand and Dickinson W. Richards) for developing a procedure that allowed cardiac catheterization. In 1929, he put himself under local anesthesia and inserted a catheter into a vein of his arm. Not knowing if the catheter might pierce a vein, he put his life at risk. Forssmann was nevertheless successful; he safely passed the catheter into his heart.
Early life
Forssmann was born in Berlin on 29 August 1904. Upon graduating from Askanisches Gymnasium [de], he entered the University of Berlin to study medicine, passing the State Examination in 1929.
Career
He hypothesized that a catheter could be inserted directly into the heart, for such applications as directly delivering drugs, injecting radiopaque dyes, or measuring blood pressure. The fear at the time was that such an intrusion into the heart would be fatal. To prove his point, he decided to try the experiment on himself.
In 1929, while working in Eberswalde, he performed the first human cardiac catheterization. He ignored his department chief and persuaded the operating-room nurse in charge of the sterile supplies, Gerda Ditzen, to assist him. She agreed, but only on the promise that he would do it on her rather than on himself. However, Forssmann tricked her by restraining her to the operating table and pretending to locally anaesthetise and cut her arm whilst actually doing it on himself. He anesthetized his own lower arm in the cubital region and inserted a urinary catheter into his antecubital vein, threading it partly along before releasing Ditzen (who at this point realised the catheter was not in her arm) and telling her to call the X-ray department. They walked some distance to the X-ray department on the floor below where under the guidance of a fluoroscope he advanced the catheter the full 60 cm into his right ventricular cavity. This was then recorded on X-ray film showing the catheter lying in his right atrium.
The head clinician at Eberswalde, although initially very annoyed, recognized Werner's discovery when shown the X-rays; he allowed Forssmann to carry out another catheterization on a terminally ill woman whose condition improved after being given drugs in this way. An unpaid position was created for Forssmann at the Berliner Charité Hospital, working under Ferdinand Sauerbruch, although once Sauerbruch saw his paper, he was dismissed for continuing without his approval. Sauerbruch commented, "You certainly can't begin surgery in that manner". Facing such disciplinary action for self-experimentation, he was initially forced to leave the Charité, but was later reinstated until again being forced to leave in 1932 for not meeting scientific expectations. His surgical skills were noted, however, and he was recommended to another hospital where he worked for a while before leaving in 1933 after marrying Dr. Elsbet Engel, a specialist in urology there. Finding it difficult to get a job with his reputation, he quit cardiology and took up urology. He then went on to study urology under Karl Heusch at the Rudolf Virchow Hospital [de] in Berlin. Later, he was appointed chief of the surgical clinic at both the City Hospital at Dresden-Friedrichstadt and the Robert Koch Hospital [de] in Berlin.
From 1932 to 1945, he was a member of the Nazi Party. At the start of World War II, he became a medical officer. In the course of his service, he rose to the rank of major, until he was captured and put into a U.S. POW camp. Upon his release in 1945, he worked as a lumberjack and then as a country medic in the Black Forest with his wife. In 1950, he began practice as a urologist in Bad Kreuznach.
During the time of his imprisonment, his paper was read by André Frédéric Cournand and Dickinson W. Richards. They developed ways of applying his technique to heart disease diagnosis and research. In 1954, he was given the Leibniz Medal of the German Academy of Sciences. In 1956, the Nobel Prize in Physiology or Medicine was awarded to Cournand, Richards, and Forßmann.
After winning the Nobel Prize, he was given the position of honorary professor of surgery and urology at the University of Mainz. In 1961, he became an honorary professor at the National University of Córdoba. In 1962, he became a member of the executive board of the German Society of Surgery. He also became a member of the American College of Chest Physicians, honorary member of the Swedish Society of Cardiology, the German Society of Urology [de], and the German Child Welfare Association.
Personal life
He and Elsbet had six children: Klaus Forßmann in 1934, Knut Forßmann in 1936, Jörg Forßmann in 1938, Wolf Forßmann in 1939 (who was first to isolate the atrial natriuretic peptide), Bernd Forßmann in 1940 (who helped develop the first clinical lithotriptor), and Renate Forßmann in 1943.
He died in Schopfheim, Germany, of heart failure on 1 June 1979. His wife died in 1993.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1880 2026-02-27 00:01:53
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2443) Dickinson W. Richards
Gist:
Life
Dickinson Richards was born in New Jersey, the son of a lawyer. After liberal arts studies at Yale University, Richards studied medicine at Columbia University in New York, becoming a medical doctor in 1923. Following work at Presbyterian Hospital in New York and elsewhere, in 1931 he began a prolonged collaboration with André Cournand at Bellevue Hospital and Columbia University. Richards was married and had four children.
Work
Even though Werner Forssmann succeeded in inserting a catheter into his own heart in 1929, there was great hesitance about continuing this type of research. Nonetheless, beginning in 1941 Dickinson Richards and André Cournand published a series of studies that established use of cardiac catheterization, among other things, to introduce contrast fluid for X-ray images and to measure pressure and oxygen content. Because it was possible to reach the upper chambers of the heart, blood pressure and the blood’s oxygen content could be measured on the way from the heart to the lungs, which was impossible before.
Summary
Dickinson Woodruff Richards (born Oct. 30, 1895, Orange, N.J., U.S.—died Feb. 23, 1973, Lakeville, Conn.) was an American physiologist who shared the Nobel Prize for Physiology or Medicine in 1956 with Werner Forssmann and André F. Cournand. Cournand and Richards adapted Forssmann’s technique of using a flexible tube (catheter), conducted from an elbow vein to the heart, as a probe to investigate the heart.
Richards received an A.B. degree from Yale University in 1917 and later studied at Columbia University’s College of Physicians and Surgeons (M.A., 1922; M.D., 1923). After a hospital internship and a brief study in England, he returned to Columbia University in 1928 and taught there from 1947 to 1961. From 1945 to 1961 he worked at Bellevue Hospital, New York City, where he met Cournand. Their use and perfection of Forssmann’s method, known as cardiac catheterization, permitted them to measure blood pressure and other conditions inside the heart.
Details
Dickinson Woodruff Richards Jr. (October 30, 1895 – February 23, 1973) was an American physician and physiologist. He was a co-recipient of the Nobel Prize in Physiology or Medicine in 1956 with André Cournand and Werner Forssmann for the development of cardiac catheterization and the characterisation of a number of cardiac diseases.
Early life
Richards was born in Orange, New Jersey. He was educated at the Hotchkiss School in Connecticut, and entered Yale University in 1913. At Yale he studied English and Greek, graduating in 1917 as a member of the senior society Scroll and Key.
Career
He joined the United States Army in 1917, and became an artillery instructor. He served from 1918 to 1919 as an artillery officer in France.
When he returned to the United States, Richards attended Columbia University College of Physicians and Surgeons, graduating with an M.A. in 1922 and his M.D. degree in 1923. He was on the staff of the Presbyterian Hospital in New York until 1927, when he went to England to work at the National Institute for Medical Research in London, under Sir Henry Dale, on the control of circulation in the liver.
In 1928, Richards returned to the Presbyterian Hospital and began his research on pulmonary and circulatory physiology, working under Professor Lawrence Henderson of Harvard. He began collaborations with André Cournand at Bellevue Hospital, New York, working on pulmonary function. Initially their research focussed on methods to study pulmonary function in patients with pulmonary disease.
Their next area of research was the development of a technique for catheterization of the heart. Using this technique they were able to study and characterise traumatic shock, the physiology of heart failure. They measured the effects of cardiac drugs and described various forms of dysfunction in chronic cardiac diseases and pulmonary diseases and their treatment, and developed techniques for the diagnosis of congenital heart diseases. For this work, Richards, Cournand, and Werner Forssmann were awarded the Nobel Prize for Physiology or Medicine for 1956.
In 1945 Richards moved his lab to Bellevue Hospital, New York. In 1947 he was made the Lambert Professor of Medicine at Columbia University, where he had taught since 1925. During his career he also served as an advisor to Merck Sharp and Dohme Company, and edited the Merck Manual. Richards retired from his positions at Bellevue and Columbia in 1961.
Global policy
He was one of the signatories of the agreement to convene a convention for drafting a world constitution. As a result, for the first time in human history, a World Constituent Assembly convened to draft and adopt the Constitution for the Federation of Earth.
Honor
Richards received many other honors, including the John Phillips Memorial Award of the American College of Physicians in 1960, the Chevalier de la Legion d'Honneur in 1963, the Trudeau Medal in 1968, and the Kober Medal of the Association of American Physicians in 1970.
He died in Lakeville, Connecticut and his wife Constance in 1990.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1881 2026-02-28 00:02:59
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2444) Willis Lamb
Gist:
Work
According to Niels Bohr’s atomic model, a photon is emitted when an electron descends to a lower energy level. This results in a spectrum with lines corresponding to the different energy levels of different atoms. It appeared that the lines were divided into several lines close to one another, which Paul Dirac tried to explain in a theory. However, in 1947 Willis Lamb used precise measurements to establish what became known as the Lamb shift: what ought to have been a single energy level in the hydrogen atom according to Dirac’s theory actually was two nearby levels with a small difference in energy.
Summary
Willis Eugene Lamb, Jr. (born July 12, 1913, Los Angeles, Calif., U.S.—died May 15, 2008, Tucson, Ariz.) was an American physicist and corecipient, with Polykarp Kusch, of the 1955 Nobel Prize for Physics for experimental work that spurred refinements in the quantum theories of electromagnetic phenomena.
Lamb joined the faculty of Columbia University, New York City, in 1938 and worked in the Radiation Laboratory there during World War II. Though the quantum mechanics of P.A.M. Dirac had predicted the hyperfine structure of the lines that appear in the spectrum (dispersed light, as by a prism), Lamb applied new methods to measure the lines and in 1947 found their positions to be slightly different from what had been predicted. While a professor of physics (1951–56) at Stanford University, California, Lamb devised microwave techniques for examining the hyperfine structure of the spectral lines of helium. He was a professor of theoretical physics at the University of Oxford until 1962, when he was appointed a professor of physics at Yale University. In 1974 he became a professor of physics and optical sciences at the University of Arizona; he retired as professor emeritus in 2002.
Details
Willis Eugene Lamb Jr. (July 12, 1913 – May 15, 2008) was an American physicist who shared the 1955 Nobel Prize in Physics with Polykarp Kusch "for his discoveries concerning the fine structure of the hydrogen spectrum". Lamb was able to precisely determine a surprising shift in electron energies in a hydrogen atom, known as the Lamb shift. He was a professor at the University of Arizona College of Optical Sciences.
Biography
Lamb was born in Los Angeles, California, and attended Los Angeles High School. First admitted in 1930, he received a Bachelor of Science in chemistry from the University of California, Berkeley in 1934. For theoretical work on scattering of neutrons by a crystal, guided by J. Robert Oppenheimer, he received the Ph.D. in physics in 1938. Because of limited computational methods available at the time, this research narrowly missed revealing the Mössbauer Effect, 19 years before its recognition by Rudolf Mössbauer. He worked on nuclear theory, laser physics, and verifying quantum mechanics.
Lamb was a physics professor at Stanford from 1951 to 1956. He was the Wykeham Professor of Physics at the University of Oxford from 1956 to 1962, and also taught at Yale, Columbia and the University of Arizona. He was elected a Fellow of the American Academy of Arts and Sciences in 1963. In 2000, The Optical Society elected him an Honorary member.
Lamb is remembered as a "rare theorist turned experimentalist" by D. Kaiser.
Quantum physics
In addition to his crucial and famous contribution to quantum electrodynamics via the Lamb shift, in the latter part of his career he paid increasing attention to the field of quantum measurements. In one of his writings Lamb stated that "most people who use quantum mechanics have little need to know much about the interpretation of the subject." Lamb was also openly critical of many of the interpretational trends on quantum mechanics and of the use of the term photon.
Personal
In 1939 Lamb married his first wife, Ursula Schäfer, a German student, who became a distinguished historian of Latin America (and assumed his last name). After her death in 1996, he married physicist Bruria Kaufman in 1996, whom he later divorced. In 2008 he married Elsie Wattson.
Lamb died on May 15, 2008, at the age of 94, due to complications of a gallstone disorder.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1882 2026-03-01 00:02:42
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2445) Polykarp Kusch
Gist:
Work
In certain respects, electrons and atoms behave as if they were rotating charged particles that generate a magnetic field. Their magnetic moment provides a way of measuring how strongly they are affected by a magnetic field. Drawing upon quantum mechanics and relativity theory, Paul Dirac developed a theory for the interaction of charged particles with electromagnetic fields. Through precise measurements, Polykarp Kusch showed in 1947 that the electron’s magnetic moment was somewhat greater than the theory predicted. This became important for the further development of quantum electrodynamics.
Summary
Polykarp Kusch (born Jan. 26, 1911, Blankenburg, Ger.—died March 20, 1993, Dallas, Texas, U.S.) was a German-American physicist who, with Willis E. Lamb, Jr., was awarded the Nobel Prize for Physics in 1955 for his accurate determination that the magnetic moment of the electron is greater than its theoretical value, thus leading to reconsideration of and innovations in quantum electrodynamics.
Kusch was brought to the United States in 1912 and became a citizen in 1922. In 1937, at Columbia University, he worked with the physicist Isidor I. Rabi on studies of the effects of magnetic fields on beams of atoms. He spent the wartime years in research on radar and returned to Columbia in 1946 as professor of physics, a position he held until 1972. Among other posts held by Kusch at Columbia were department chairman (1949–52, 1960–63), director of the radiation laboratory (1952–60), and academic vice president and provost (1969–72). In 1972 he took a position as professor at the University of Texas, Dallas, where he remained until his retirement in 1982.
In 1947, through precise atomic beam studies, Kusch demonstrated that the magnetic properties of the electron were not in agreement with existing theories. Subsequently, he made accurate measurements of the magnetic moment of the electron and its behaviour in hydrogen. In work characterized by great accuracy and reliability, he measured numerous atomic, molecular, and nuclear properties by radio-frequency beam techniques.
Details
Polykarp Kusch (January 26, 1911 – March 20, 1993) was a German-American physicist who shared the 1955 Nobel Prize in Physics with Willis Eugene Lamb for his accurate determination that the electron magnetic moment was greater than its theoretical value, thus leading to reconsideration of and innovations in quantum electrodynamics.
Early life and education
Kusch was born in Blankenburg, Germany to John Mathias Kusch, a Lutheran missionary, and his wife, Henrietta van der Haas. In 1912, Kusch and his family had emigrated to the United States, where by 1922 he became a naturalized citizen. After graduating from grade school in the Midwest, Kusch attended Case Institute of Technology in Cleveland, Ohio (now known as Case Western Reserve University), where he majored in physics. After graduating from the Case Western Reserve University with bachelor of science degree in 1931, Kusch joined University of Illinois at Urbana–Champaign, from where he received his master's degree in 1933. He continued his education at the same alma mater, studying for his Ph.D. under mentorship from F. Wheeler Loomis and after defending his thesis titled "The Molecular Spectrum of Caesium and Rubidium", graduated from it in 1936. In 1935, prior to moving to the University of Minnesota, Kusch married his girlfriend, Edith Starr McRoberts. Together, they had three daughters.
Career
Kusch then moved to New York City, where from 1937 and until his departure for the newly founded University of Texas at Dallas, he spent much of his career as a professor at Columbia University, and served as the university's provost for several years. He worked on molecular beam resonance studies under I. I. Rabi, then discovered the electron anomalous magnetic moment. Many measurements of magnetic moments and hyperfine structure followed. He expanded into chemical physics and continued to publish research on molecular beams. During his tenure at Columbia, he was the doctoral supervisor for Gordon Gould, the inventor of the laser.
Kusch was a fellow of the American Physical Society since 1940 and of the American Academy of Arts and Sciences since 1959. He was elected a member of the National Academy of Sciences in 1956. In 1967, he was elected to the American Philosophical Society.
Kusch's wife Edith died in 1959, and in the following year he married Betty Pezzoni. They had two daughters. Kusch House, a residential dormitory for undergraduate students at Case Western Reserve University in Cleveland, Ohio on the South Campus, is named after Kusch. It is located on Carlton Road in Cleveland Heights. The University of Texas at Dallas has a Polykarp Kusch Auditorium with a plaque.
Kusch died on March 20, 1993, aged 82. His widow Betty died in 2003, aged 77.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1883 2026-03-02 00:05:46
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2446) Vincent du Vigneaud
Gist:
Work
The element sulfur plays an important role in some of the chemical compounds and processes that are the basis of all life. Vincent du Vigneaud studied sulfurous compounds, including oxytocin, a hormone that among other things plays a role in sexual intimacy and reproduction among people and mammals. In 1953 du Vigneaud succeeded in isolating the substance and determining its chemical composition. It became the first peptide hormone to have its sequence of amino acids determined. He also succeeded in producing oxytocin by artificial means.
Summary
Vincent du Vigneaud (born May 18, 1901, Chicago, Illinois, U.S.—died December 11, 1978, White Plains, New York) was an American biochemist and winner of the Nobel Prize for Chemistry in 1955 for the isolation and synthesis of two pituitary hormones: vasopressin, which acts on the muscles of the blood vessels to cause elevation of blood pressure; and oxytocin, the principal agent causing contraction of the uterus and secretion of milk.
Du Vigneaud studied at the University of Illinois at Urbana-Champaign, took a doctorate from the University of Rochester, New York (1927), and then studied at Johns Hopkins University, Baltimore, the Kaiser Wilhelm Institute, Berlin, and the University of Edinburgh. He headed the biochemistry department of the George Washington University Medical School, Washington, D.C. (1932–38), and was professor and head of the department of biochemistry at the Cornell University Medical College, New York City (1938–67), and professor of chemistry at Cornell University, Ithaca, New York (1967–75).
Du Vigneaud and his staff at Cornell helped identify the chemical structure of the hormone insulin in the late 1930s, and in the early 1940s they established the structure of the sulfur-bearing vitamin biotin. Later that decade, they isolated vasopressin and oxytocin and analyzed both those hormones’ chemical structure. Du Vigneaud found that the oxytocin molecule contains only eight different amino acids (nine amino acids in total, whereby a disulfide bond forms a link between two cysteines), in contrast to the hundreds of amino acids most other proteins contain. In 1953 he was able to synthesize oxytocin, becoming the first to achieve the synthesis of a protein hormone. In 1946 du Vigneaud and his colleagues at Cornell achieved another breakthrough, the synthesis of penicillin.
Details
Vincent du Vigneaud (May 18, 1901 – December 11, 1978) was an American biochemist. He was recipient of the 1955 Nobel Prize in Chemistry "for his work on biochemically important sulphur compounds, especially for the first synthesis of a polypeptide hormone," a reference to his work on the peptide hormone oxytocin.
Biography
Vincent du Vigneaud was born in Chicago in 1901. Of French descent, he was the son of inventor and mechanic Alfred du Vigneaud and Mary Theresa. He studied at the Schurz High School and completed secondary education in 1918. His interest in sulfur began when he entered high school and his new friends invited him to run chemical experiments on explosives using sulfur. During World War I, senior students were made to work on farms, and du Vigneaud worked near Caledonia, Illinois. There he became an expert in milking cows, which inspired him to become a farmer. However, his elder sister, Beatrice, persuaded him to take up chemistry at the University of Illinois at Urbana-Champaign, after which he enrolled in the chemical engineering course. He later recalled:
I found during the first year that it was chemistry rather than engineering that appealed to me most. I switched to a major in chemistry, since I was deeply impressed by the senior student's work, especially in organic chemistry. I also found that I was most interested in those aspects of organic chemistry that had to do with medical substances and began to develop an interest in biochemistry.
His interest was aroused by lectures of Carl Shipp Marvel and Howard B. Lewis, whom he remembered as being 'extremely enthusiastic about sulfur." With little support from the family, he found odd jobs to support himself. After receiving his MS in 1924 he joined DuPont.
He married Zella Zon Ford, whom he met on June 12, 1924, while working as a waiter during his university course. During the fall of 1924, Marvel found him a job as an assistant biochemist at the Philadelphia General Hospital that helped him to teach clinical chemistry at the Graduate School of Medicine, University of Pennsylvania. Marvel would pay for the trip to Pennsylvania in exchange for du Vigneaud's preparation of 10 pounds of cupferron. Resuming his academic career in 1925, du Vigneaud joined the group of John R. Murlin at the University of Rochester for his PhD thesis. He graduated in 1927 with his work The Sulfur of Insulin.
After a post-doctoral position with John Jacob Abel at Johns Hopkins University Medical School (1927–1928), he traveled to Europe as a National Research Council Fellow in 1928–1929, where he worked with Max Bergmann and Leonidas Zervas at the Kaiser Wilhelm Institute for Leather Research in Dresden, and with George Barger at the University of Edinburgh Medical School. He then returned to the University of Illinois as a professor.
In 1932, he started working at the George Washington University Medical School in Washington, D.C., and in 1938, he attended the Cornell Medical College in New York City, where he stayed until his emeritation in 1967. Following retirement, he held a position at Cornell University in Ithaca, New York.
In 1974, du Vigneaud had a stroke which forced his retirement. He died in 1978, one year after his wife's death in 1977.
Scientific contributions
Du Vigneaud's career was characterized by an interest in sulfur-containing peptides, proteins, and especially peptide hormones. Even before his Nobel-Prize-winning work on elucidating and synthesizing oxytocin and vasopressin via manipulating the AVP gene, he had established a reputation from his research on insulin, biotin, transmethylation, and penicillin.
He also carried out a series of structure-activity relationships for oxytocin and vasopressin, perhaps the first of their type for peptides. That work culminated in the publication of a book entitled A Trail of Research in Sulphur Chemistry and Metabolism and Related Fields.
Honours
Du Vigneaud joined Alpha Chi Sigma while at the University of Illinois in 1930. He was elected to the United States National Academy of Sciences and the American Philosophical Society in 1944, and the American Academy of Arts and Sciences in 1948. He received the 1955 Nobel Prize in Chemistry "for his work on biochemically important sulphur compounds, especially for the first synthesis of a polypeptide hormone," a reference to his work on the peptide hormone oxytocin.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1884 2026-03-03 00:05:27
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2447) Hugo Theorell
Gist:
Life
Hugo Theorell was born in Linköping, Sweden. His father worked as a regimental medical officer. Theorell studied medicine at Karolinska Institutet, Stockholm, receiving his degree in 1930. After receiving a Rockefeller Fellowship, he worked with Otto Warburg in Berlin-Dahlem from 1933 to 1935, where he conducted some of his most important work. He became a professor at Sweden's Uppsala University in 1937 and then at Karolinska Institutet in 1959. Theorell was married to concert pianist Margit Alenius and had three sons.
Work
Catalysts are substances that speed up chemical reactions without influencing the end products. Enzymes are catalysts active in biological processes. Hugo Theorell and colleagues investigated how enzymes that promote oxidation reactions are constructed and function. A breakthrough came in 1935 when he demonstrated how a yellow-colored enzyme in yeast had two parts, both of which were crucial to its function. He also explained how iron atoms in many enzymes have an important function in transporting electrons.
Summary
Axel Hugo Teodor Theorell (born July 6, 1903, Linköping, Sweden—died Aug. 15, 1982, Stockholm) was a Swedish biochemist whose study of enzymes that facilitate oxidation reactions in living cells contributed to the understanding of enzyme action and led to the discovery of the ways in which nutrients are used by organisms in the presence of oxygen to produce usable energy. Theorell won the Nobel Prize for Physiology or Medicine in 1955.
While serving as an assistant professor of biochemistry at Uppsala University (1932–33; 1935–36), Theorell was the first to isolate crystalline myoglobin, an oxygen-carrying protein found in red muscle (1932). At the Kaiser Wilhelm Institute (now Max Planck Institute), Berlin (1933–35), he worked with Otto Warburg in isolating from yeast a pure sample of the “old yellow enzyme,” which is instrumental in the oxidative interconversion of sugars by the cell. Theorell found that the enzyme is composed of two parts: a nonprotein coenzyme—the yellow riboflavine (vitamin B2) phosphate—and a protein apoenzyme. His discovery (1934) that the coenzyme actively facilitates oxidation of the sugar glucose by binding a hydrogen atom at a specific site on the riboflavin molecule marked the first time that the effect of an enzyme was attributed to the chemical activity of specific atoms.
As director of the biochemical department of the Nobel Medical Institute, Stockholm (1937–70), Theorell studied the oxidative enzyme cytochrome c, determining the precise nature of the chemical linkage between the iron-bearing, nonprotein porphyrin portion and the apoenzyme. His investigation of the hydrogen-transfer enzyme, alcohol dehydrogenase, led to the development of sensitive blood tests that have found wide application in the determination of legal definitions of intoxication. Besides the Nobel Prize, Theorell received a number of awards and honours. He also served as president of the Swedish Royal Academy of Science and International Union of Biochemistry.
Details
Axel Hugo Theodor Theorell (6 July 1903 – 15 August 1982) was a Swedish scientist and Nobel Prize laureate in medicine.
Life
He was born in Linköping as the son of Thure Theorell and his wife Armida Bill. Theorell went to Secondary School at Katedralskolan in Linköping and passed his examination there on 23 May 1921. In September, he began to study medicine at the Karolinska Institute and in 1924 he graduated as a Bachelor of Medicine. He then spent three months studying bacteriology at the Pasteur Institute in Paris under Professor Albert Calmette. In 1930 he obtained his M.D. degree with a theory on the lipids of the blood plasma, and was appointed professor in physiological chemistry at the Karolinska Institute.
Theorell, who dedicated his entire career to enzyme research, received the Nobel Prize in Physiology or Medicine in 1955 for discovering oxidoreductase enzymes and their effects. His contribution also consisted of the theory of the toxic effects of sodium fluoride on the cofactors of crucial human enzymes. In 1936 he was appointed Head of the newly established Biochemical Department of the Nobel Medical Institute, the first researcher related to the Institute to be awarded a Nobel Prize. His work had led to pioneering progress on alcohol dehydrogenases, enzymes that break down alcohol in the liver and other tissues. He received honorary degrees at universities in France, Belgium, Brazil and the United States. He was a member of the American Academy of Arts and Sciences and the United States National Academy of Sciences, and an International Member of the American Philosophical Society.
Theorell died in Stockholm and is interred in Norra begravningsplatsen (The Northern Cemetery) alongside his wife, Elin Margit Elisabeth (née Alenius) Theorell, a distinguished pianist and harpsichordist who died in 2002.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1885 2026-03-04 00:02:57
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2448) Max Born
Gist:
Life
Max Born was born in Breslau, Germany (now Wroclaw, Poland), where his father was a professor of anatomy. Born studied at universities in Breslau, Heidelberg, Zurich and Göttingen, where he received his doctorate in 1906. After serving in the army during the First World War, he became a professor at the University of Frankfurt-on-Main in 1919 and at the University of Göttingen in 1921. After the Nazis seized power in 1933, he moved to Cambridge in United Kingdom. Beginning in 1936, he served as a professor at the University of Edinburgh. Max Born was married and had three children.
Work
In Niels Bohr’s theory of the atom, electrons absorb and emit radiation of fixed wavelengths when jumping between orbits around a nucleus. The theory provided a good description of the spectrum created by the hydrogen atom, but needed to be developed to suit more complicated atoms and molecules. Following Werner Heisenberg’s initial work around 1925, Max Born contributed to the further development of quantum mechanics. He also proved that Schrödinger’s wave equation could be interpreted as giving statistical (rather than exact) predictions of variables.
Summary
Max Born (born Dec. 11, 1882, Breslau, Ger. [now Wrocław, Pol.]—died Jan. 5, 1970, Göttingen, W.Ger.) was a German physicist who shared the Nobel Prize for Physics in 1954 with Walther Bothe for his probabilistic interpretation of quantum mechanics.
Born came from an upper-middle-class, assimilated, Jewish family. At first he was considered too frail to attend public school, so he was tutored at home before being allowed to attend the König Wilhelm Gymnasium in Breslau. Thereafter he continued his studies in physics and mathematics at universities in Breslau, Heidelberg, Zürich, and Göttingen. At the University of Göttingen he wrote his dissertation (1906), on the stability of elastic wires and tapes, under the direction of the mathematician Felix Klein, for which he was awarded a doctorate in 1907.
After brief service in the army and a stay at the University of Cambridge, where he worked with physicists Joseph Larmor and J.J. Thomson, Born returned to Breslau for the academic year 1908–09 and began an extensive study of Albert Einstein’s theory of special relativity. On the strength of his papers in this field, Born was invited back to Göttingen as an assistant to the mathematical physicist Hermann Minkowski. In 1912 Born met Hedwig Ehrenberg, whom he married a year later. Three children, two girls and a boy, were born from the union. It was a troubled relationship, and Born and his wife often lived apart.
In 1915 Born accepted a professorship to assist physicist Max Planck at the University of Berlin, but World War I intervened and he was drafted into the German army. Nonetheless, while an officer in the army, he found time to publish his first book, Dynamik der Kristallgitter (1915; Dynamics of Crystal Lattices).
In 1919 Born was appointed to a full professorship at the University of Frankfurt am Main, and in 1921 he accepted the position of professor of theoretical physics at the University of Göttingen. James Franck had been appointed professor of experimental physics at Göttingen the previous year. The two of them made the University of Göttingen one of the most important centres for the study of atomic and molecular phenomena. A measure of Born’s influence can be gauged by the students and assistants who came to work with him—among them, Wolfgang Pauli, Werner Heisenberg, Pascual Jordan, Enrico Fermi, Fritz London, P.A.M. Dirac, Victor Weisskopf, J. Robert Oppenheimer, Walter Heitler, and Maria Goeppert-Mayer.
The Göttingen years were Born’s most creative and seminal. In 1912 Born and Hungarian engineer Theodore von Karman formulated the dynamics of a crystal lattice, which incorporated the symmetry properties of the lattice, allowed the imposition of quantum rules, and permitted thermal properties of the crystal to be calculated. This work was elaborated when Born was in Göttingen, and it formed the basis of the modern theory of lattice dynamics.
In 1925 Heisenberg gave Born a copy of the manuscript of his first paper on quantum mechanics, and Born immediately recognized that the mathematical entities with which Heisenberg had represented the observable physical quantities of a particle—such as its position, momentum, and energy—were matrices. Joined by Heisenberg and Jordan, Born formulated all the essential aspects of quantum mechanics in its matrix version. A short time later, Erwin Schrödinger formulated a version of quantum mechanics based on his wave equation. It was soon proved that the two formulations were mathematically equivalent. What remained unclear was the meaning of the wave function that appeared in Schrödinger’s equation. In 1926 Born submitted two papers in which he formulated the quantum mechanical description of collision processes and found that in the case of the scattering of a particle by a potential, the wave function at a particular spatiotemporal location should be interpreted as the probability amplitude of finding the particle at that specific space-time point. In 1954 he was awarded the Nobel Prize for this work.
Born remained at Göttingen until April 1933, when all Jews were dismissed from their academic posts in Germany. Born and his family went to England, where he accepted a temporary lectureship at Cambridge. In 1936 he was appointed Tait Professor of Natural Philosophy at the University of Edinburgh. He became a British citizen in 1939 and remained at Edinburgh until his retirement in 1953. The next year, he and his wife moved to Bad Pyrmont, a small spa town near Göttingen.
Details
Max Born (11 December 1882 – 5 January 1970) was a German–British theoretical physicist who was instrumental in the development of quantum mechanics. He also made contributions to solid-state physics and optics, and supervised the work of a number of notable physicists in the 1920s and 1930s. He shared the 1954 Nobel Prize in Physics with Walther Bothe "for his fundamental research in quantum mechanics, especially for his statistical interpretation of the wavefunction."
Born entered the University of Göttingen in 1904, where he met the three renowned mathematicians Felix Klein, David Hilbert, and Hermann Minkowski. He wrote his Ph.D. thesis on the subject of the stability of elastic wires and tapes, winning the university's Philosophy Faculty Prize. In 1905, he began researching special relativity with Minkowski, and subsequently wrote his habilitation thesis on the Thomson model of the atom. A chance meeting with Fritz Haber in Berlin in 1918 led to discussion of how an ionic compound is formed when a metal reacts with a halogen, which is now known as the Born–Haber cycle.
During World War I, Born was originally placed as a radio operator, but his specialist knowledge led to his being moved to research duties on sound ranging. In 1921 Born returned to Göttingen, where he arranged another chair for his long-time friend and colleague James Franck. Under Born, Göttingen became one of the world's foremost centres for physics. In 1925, Born and Werner Heisenberg formulated the matrix mechanics representation of quantum mechanics. The following year, he formulated the now-standard interpretation of the probability density function for ψ*ψ in the Schrödinger equation, for which he was awarded the Nobel Prize in 1954.
His influence extended far beyond his own research: Max Delbrück, Siegfried Flügge, Friedrich Hund, Pascual Jordan, Maria Goeppert Mayer, Lothar Nordheim, Robert Oppenheimer, and Victor Weisskopf all received their Ph.D. degrees under Born at Göttingen, and his assistants included Enrico Fermi, Werner Heisenberg, Gerhard Herzberg, Friedrich Hund, Wolfgang Pauli, Léon Rosenfeld, Edward Teller, and Eugene Wigner.
In January 1933, when the Nazi Party came to power in Germany, Born, who was born into a Jewish family, was suspended from his professorship at the University of Göttingen. He emigrated to the United Kingdom, where he took a job at St John's College, Cambridge, and wrote a popular science book, The Restless Universe, as well as Atomic Physics, which soon became a standard textbook. In October 1936, he was appointed Tait Professor of Natural Philosophy at the University of Edinburgh, where, working with German-born assistants E. Walter Kellermann and Klaus Fuchs, he continued his research into physics. He became a naturalised British subject on 31 August 1939, one day before World War II broke out in Europe. He remained in Edinburgh until 1952, when he retired to Bad Pyrmont, West Germany, and died in a hospital in Göttingen on 5 January 1970.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1886 2026-03-05 00:02:53
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2449) Walther Bothe
Gist:
Work
In a counter tube, particles passing through the tube generate an electric pulse. In 1925 Walter Bothe connected two counter tubes together so that only simultaneous passages were registered. This meant that either the passages were caused by particles that originated from the same event or by a particle that moved so fast that the time for movement between the tubes was negligible. Bothe used the method to show that energy is conserved in impacts between particles and photons and to study cosmic radiation.
Summary
Walther Bothe (born Jan. 8, 1891, Oranienburg, Ger.—died Feb. 8, 1957, Heidelberg, W.Ger.) was a German physicist who shared the Nobel Prize for Physics in 1954 with Max Born for his invention of a new method of detecting subatomic particles and for other resulting discoveries.
Bothe taught at the universities of Berlin (1920–31), Giessen (1931–34), and Heidelberg (1934–57). In 1925 he and Hans Geiger used two Geiger counters to gather data on the Compton effect—the dependence of the increase in the wavelength of a beam of X rays upon the angle through which the beam is scattered as a result of collision with electrons. Their experiments, which simultaneously measured the energies and directions of single photons and electrons emerging from individual collisions, refuted a statistical interpretation of the Compton effect and definitely established the particle nature of electromagnetic radiation.
With the astronomer Werner Kolhörster, Bothe again applied this coincidence-counting method in 1929 and found that cosmic rays are not composed exclusively of gamma rays, as was previously believed. In 1930 Bothe discovered an unusual radiation emitted by beryllium when it is bombarded with alpha particles. This radiation was later identified by Sir James Chadwick as the neutron.
During World War II Bothe was one of the leaders of German research on nuclear energy. He was responsible for the planning and building of Germany’s first cyclotron, which was completed in 1943.
Details
Walther Wilhelm Georg Bothe (8 January 1891 – 8 February 1957) was a German experimental physicist who shared the 1954 Nobel Prize in Physics with Max Born "for the coincidence method and his discoveries made therewith."
Bothe served in the military during World War I from 1914, and he was a prisoner of war of the Russians, returning to Germany in 1920. Upon his return to the laboratory, he developed and applied coincidence circuits to the study of nuclear reactions, such as the Compton effect, cosmic rays, and the wave–particle duality of radiation.
In 1930, Bothe became Full Professor and Director of the Physics Department at the University of Giessen. In 1932, he became Director of the Physical and Radiological Institute at the University of Heidelberg; he was driven out of this position by elements of the Deutsche Physik movement. To preclude his emigration from Germany, he was appointed Director of the Physics Institute of the Kaiser Wilhelm Institute for Medical Research in Heidelberg. There, he built the first operational cyclotron in Germany. Furthermore, he became a principal in the German nuclear energy project, also known as Uranverein, which was started in 1939 under the supervision of the Army Ordnance Office.
In 1946, in addition to his directorship of the Physics Institute at the KWImf, Bothe was reinstated as a professor at the University of Heidelberg. From 1956 to 1957, he was a member of the Nuclear Physics Working Group in Germany.
In the year after Bothe's death, his Physics Institute at the KWImF was elevated to the status of a new institute under the Max Planck Society and it then became the Max Planck Institute for Nuclear Physics. Its main building was later named Bothe laboratory.
Education
Walther Wilhelm Georg Bothe was born on 8 January 1891 in Oranienburg, Germany, the son of Friedrich Bothe and Charlotte Hartung.
From 1908 to 1912, Bothe studied at the University of Berlin. In 1913, he became Max Planck's teaching assistant. He received his Ph.D. under Planck the following year.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1887 2026-03-06 00:05:02
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2450) Linus Pauling
Gist:
Life
Linus Pauling was born in Portland, Oregon, in the United States. His family came from a line of Prussian farmers, and Pauling's father worked as a pharmaceuticals salesman, among other things. After first studying at Oregon State University in Corvallis, Oregon, Pauling earned his PhD from the California Institute of Technology in Pasadena, with which he maintained ties for the rest of his career. In the 1950s, Pauling's involvement in the anti-nuclear movement led to his being labeled a suspected communist, which resulted in his passport being revoked at times. Linus and Ava Helen Pauling had four children together.
Work
The development of quantum mechanics during the 1920s had a great impact not only on the field of physics, but also on chemistry. During the 1930s Linus Pauling was among the pioneers who used quantum mechanics to understand and describe chemical bonding–that is, the way atoms join together to form molecules. Pauling worked in a broad range of areas within chemistry. For example, he worked on the structures of biologically important chemical compounds. In 1951 he published the structure of the alpha helix, which is an important basic component of many proteins.
Summary
Linus Carl Pauling (February 28, 1901 – August 19, 1994) was an American chemist and peace activist. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. New Scientist called him one of the 20 greatest scientists of all time. For his scientific work, Pauling was awarded the Nobel Prize in Chemistry in 1954. For his peace activism, he was awarded the Nobel Peace Prize in 1962. He is one of five people to have won more than one Nobel Prize. Of these, he is the only person to have been awarded two unshared Nobel Prizes, and one of two people to be awarded Nobel Prizes in different fields, the other being Marie Skłodowska-Curie.
Pauling was one of the founders of the fields of quantum chemistry and molecular biology. His contributions to the theory of the chemical bond include the concept of orbital hybridisation and the first accurate scale of electronegativities of the elements. Pauling also worked on the structures of biological molecules, and showed the importance of the alpha helix and beta sheet in protein secondary structure. Pauling's approach combined methods and results from X-ray crystallography, molecular model building, and quantum chemistry. His discoveries inspired the work of James Watson, Francis Crick, Rosalind Franklin, and Maurice Wilkins on the structure of DNA, which in turn made it possible for geneticists to crack the DNA code of all organisms.
In his later years, he promoted nuclear disarmament, as well as orthomolecular medicine, megavitamin therapy, and dietary supplements, especially ascorbic acid (commonly known as Vitamin C). None of his ideas concerning the medical usefulness of large doses of vitamins have gained much acceptance in the mainstream scientific community. He was married to the American human rights activist Ava Helen Pauling.
Details
Linus Pauling (born February 28, 1901, Portland, Oregon, U.S.—died August 19, 1994, Big Sur, California) was an American theoretical physical chemist who became the only person to have won two unshared Nobel Prizes. His first prize (1954) was awarded for research into the nature of the chemical bond and its use in elucidating molecular structure; the second (1962) recognized his efforts to ban the testing of nuclear weapons.
Early life and education
Pauling was the first of three children and the only son of Herman Pauling, a pharmacist, and Lucy Isabelle (Darling) Pauling, a pharmacist’s daughter. After his early education in Condon and Portland, Oregon, he attended Oregon Agricultural College (now Oregon State University), where he met Ava Helen Miller, who would later become his wife, and where he received his Bachelor of Science degree in chemical engineering summa cum laude in 1922. He then attended the California Institute of Technology (Caltech), where Roscoe G. Dickinson showed him how to determine the structures of crystals using X rays. He received his Ph.D. in 1925 for a dissertation derived from his crystal-structure papers. Following a brief period as a National Research Fellow, he received a Guggenheim Fellowship to study quantum mechanics in Europe. He spent most of the 18 months at Arnold Sommerfeld’s Institute for Theoretical Physics in Munich, Germany.
Elucidation of molecular structures
After completing postdoctoral studies, Pauling returned to Caltech in 1927. There he began a long career of teaching and research. Analyzing chemical structure became the central theme of his scientific work. By using the technique of X-ray diffraction, he determined the three-dimensional arrangement of atoms in several important silicate and sulfide minerals. In 1930, during a trip to Germany, Pauling learned about electron diffraction, and upon his return to California he used this technique of scattering electrons from the nuclei of molecules to determine the structures of some important substances. This structural knowledge assisted him in developing an electronegativity scale in which he assigned a number representing a particular atom’s power of attracting electrons in a covalent bond.
To complement the experimental tool that X-ray analysis provided for exploring molecular structure, Pauling turned to quantum mechanics as a theoretical tool. For example, he used quantum mechanics to determine the equivalent strength in each of the four bonds surrounding the carbon atom. He developed a valence bond theory in which he proposed that a molecule could be described by an intermediate structure that was a resonance combination (or hybrid) of other structures. His book The Nature of the Chemical Bond, and the Structure of Molecules and Crystals (1939) provided a unified summary of his vision of structural chemistry.
The arrival of the geneticist Thomas Hunt Morgan at Caltech in the late 1920s stimulated Pauling’s interest in biological molecules, and by the mid-1930s he was performing successful magnetic studies on the protein hemoglobin. He developed further interests in protein and, together with biochemist Alfred Mirsky, Pauling published a paper in 1936 on general protein structure. In this work the authors explained that protein molecules naturally coiled into specific configurations but became “denatured” (uncoiled) and assumed some random form once certain weak bonds were broken.
On one of his trips to visit Mirsky in New York, Pauling met Karl Landsteiner, the discoverer of blood types, who became his guide into the field of immunochemistry. Pauling was fascinated by the specificity of antibody-antigen reactions, and he later developed a theory that accounted for this specificity through a unique folding of the antibody’s polypeptide chain. World War II interrupted this theoretical work, and Pauling’s focus shifted to more practical problems, including the preparation of an artificial substitute for blood serum useful to wounded soldiers and an oxygen detector useful in submarines and airplanes. J. Robert Oppenheimer asked Pauling to head the chemistry section of the Manhattan Project, but his suffering from glomerulonephritis (inflammation of the glomerular region of the kidney) prevented him from accepting this offer. For his outstanding services during the war, Pauling was later awarded the Presidential Medal for Merit.
While collaborating on a report about postwar American science, Pauling became interested in the study of sickle-cell anemia. He perceived that the sickling of cells noted in this disease might be caused by a genetic mutation in the globin portion of the blood cell’s hemoglobin. In 1949 he and his coworkers published a paper identifying the particular defect in hemoglobin’s structure that was responsible for sickle-cell anemia, which thereby made this disorder the first “molecular disease” to be discovered. At that time, Pauling’s article on the periodic law appeared in the 14th edition of Encyclopædia Britannica.
While serving as a visiting professor at the University of Oxford in 1948, Pauling returned to a problem that had intrigued him in the late 1930s—the three-dimensional structure of proteins. By folding a paper on which he had drawn a chain of linked amino acids, he discovered a cylindrical coil-like configuration, later called the alpha helix. The most significant aspect of Pauling’s structure was its determination of the number of amino acids per turn of the helix. During this same period he became interested in deoxyribonucleic acid (DNA), and early in 1953 he and protein crystallographer Robert Corey published their version of DNA’s structure, three strands twisted around each other in ropelike fashion. Shortly thereafter James Watson and Francis Crick published DNA’s correct structure, a double helix. Pauling’s efforts to modify his postulated structure had been hampered by poor X-ray photographs of DNA and by his lack of understanding of this molecule’s wet and dry forms. In 1952 he failed to visit Rosalind Franklin, working in Maurice Wilkins’s laboratory at King’s College, London, and consequently did not see her X-ray pictures of DNA. Frankin’s pictures proved to be the linchpin in allowing Watson and Crick to elucidate the actual structure. Nevertheless, Pauling was awarded the 1954 Nobel Prize for Chemistry “for his research into the nature of the chemical bond and its application to the elucidation of the structure of complex substances.”
Humanitarian activities of Linus Pauling
During the 1950s Pauling and his wife became well known to the public through their crusade to stop the atmospheric testing of nuclear weapons. In 1958 they presented an appeal for a test ban to the United Nations in the form of a document signed by 9,235 scientists from 44 countries. Pauling’s sentiments were also promulgated through his book No More War! (1958), a passionate analysis of the implications of nuclear war for humanity. In 1960 he was called upon to defend his actions regarding a test ban before a congressional subcommittee. By refusing to reveal the names of those who had helped him collect signatures, he risked going to jail—a stand initially condemned but later widely admired. His work on behalf of world peace was recognized with the 1962 Nobel Prize for Peace awarded on October 10, 1963, the date that the Nuclear Test Ban Treaty went into effect.
Pauling’s Peace Prize generated such antagonism from Caltech administrators that he left the institute in 1963. He became a staff member at the Center for the Study of Democratic Institutions in Santa Barbara, California, where his humanitarian work was encouraged. Although he was able to develop a new model of the atomic nucleus while working at the Center, his desire to perform more experimental research led him to a research professorship at the University of California in San Diego in 1967. There he published a paper on orthomolecular psychiatry that explained how mental health could be achieved by manipulating substances normally present in the body. Two years later he accepted a post at Stanford University, where he worked until 1972.
Later years
While at San Diego and Stanford, Pauling’s scientific interests centred on a particular molecule—ascorbic acid (vitamin C). He examined the published reports about this vitamin and concluded that, when taken in large enough quantities (megadoses), it would help the body fight off colds and other diseases. The outcome of his research was the book Vitamin C and the Common Cold (1970), which became a best-seller. Pauling’s interest in vitamin C in particular and orthomolecular medicine in general led, in 1973, to his founding an institute that eventually bore his name—the Linus Pauling Institute of Science and Medicine. During his tenure at this institute, he became embroiled in controversies about the relative benefits and risks of ingesting megadoses of various vitamins. The controversy intensified when he advocated vitamin C’s usefulness in the treatment of cancer. Pauling and his collaborator, the Scottish physician Ewan Cameron, published their views in Cancer and Vitamin C (1979). Their ideas were subjected to experimental animal studies funded by the institute. While these studies supported their ideas, investigations at the Mayo Clinic involving human cancer patients did not corroborate Pauling’s results.
Although he continued to receive recognition for his earlier accomplishments, Pauling’s later work provoked considerable skepticism and controversy. His cluster model of the atomic nucleus was rejected by physicists, his interpretation of the newly discovered quasicrystals received little support, and his ideas on vitamin C were rejected by the medical establishment. In an effort to raise money to support his increasingly troubled institute, Pauling published How to Live Longer and Feel Better (1986), but the book failed to become the success that he and his associates had anticipated.
Both Pauling and his wife developed cancer. Ava Helen Pauling died of stomach cancer in 1981. Ten years later Pauling discovered that he had prostate cancer. Although he underwent surgery and other treatments, the cancer eventually spread to his liver. He died at his ranch on the Big Sur coast of California.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1888 2026-03-07 00:03:06
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2451) John Franklin Enders
Gist:
Work
Many infectious diseases are caused by viruses—very small biological particles. A virus lacks metabolism of its own and cannot multiply without infecting a living cell. For a long time the prevailing opinion was that viruses could not be cultured in a laboratory. However, in 1949 John Enders, Frederick Robbins, and Thomas Weller succeeded in culturing the virus that causes polio in human muscle and tissue in a laboratory setting. This became an important step on the road toward a vaccine against polio.
Summary
John Franklin Enders (born Feb. 10, 1897, West Hartford, Conn., U.S.—died Sept. 8, 1985, Waterford, Conn.) was an American virologist and microbiologist who, with Frederick C. Robbins and Thomas H. Weller, was awarded the Nobel Prize for Physiology or Medicine for 1954 for his part in cultivating the poliomyelitis virus in nonnervous-tissue cultures, a preliminary step to the development of the polio vaccine.
Enders was a student of English literature at Harvard University (M.A., 1922) before he turned to bacterial studies there (Ph.D., 1930). His early researches contributed new and basic knowledge to problems of tuberculosis, pneumococcal infections, and resistance to bacterial diseases. In 1929 he joined the Harvard faculty as an assistant in the department of bacteriology and immunology, later advancing to assistant professor (1935) and associate professor (1942) in the university’s medical school.
In World War II he was a civilian consultant on infectious diseases to the U.S. War Department. From 1945 to 1949 he served the U.S. Army in a like capacity, with particular work on the mumps virus and rickettsial diseases. During this period Enders, with his coworkers Weller and Robbins, began research into new methods of producing in quantity the virus of poliomyelitis. Until that time the only effective method of growing the virus had been in the nerve tissue of living monkeys, and the vaccine thus produced had been proved dangerous to humans. The Enders–Weller–Robbins method of production, achieved in test tubes using cultures of nonnerve tissue from human embryos and monkeys, led to the development of the Salk vaccine for polio in 1954. Similarly, their production in the late 1950s of a vaccine against the measles led to the development of a licensed vaccine in the United States in 1963. Much of Enders’ research on viruses was conducted at the Children’s Hospital in Boston, where he had established a laboratory in 1946.
Details
John Franklin Enders (February 10, 1897 – September 8, 1985) was an American biomedical scientist and Nobel Laureate. Enders has been called "The Father of Modern Vaccines."
Life and education
Enders was born in West Hartford, Connecticut on February 10, 1897. His father, John Ostrom Enders, was CEO of the Hartford National Bank and left him a fortune of $19 million upon his death. He attended the Noah Webster School in Hartford, and graduated from St. Paul's School in Concord, New Hampshire in 1915. After attending Yale University a short time, he joined the United States Army Air Corps in 1918 as a flight instructor and a lieutenant.
After returning from World War I, he graduated from Yale, where he was a member of Scroll and Key as well as Delta Kappa Epsilon. He went into real estate in 1922, and tried several careers before choosing the biomedical field with a focus on infectious diseases, gaining a PhD at Harvard in 1930. He later joined the faculty at Children's Hospital Boston.
Enders died at his summer home in Waterford, Connecticut, aged 88, on 8 September 1985. His wife died in 2000.
Biomedical career
In 1949, Enders, Thomas Huckle Weller, and Frederick Chapman Robbins reported successful in vitro culture of an animal virus—poliovirus. The three received the 1954 Nobel Prize in Physiology or Medicine "for their discovery of the ability of poliomyelitis viruses to grow in cultures of various types of tissue".
Meanwhile, Jonas Salk applied the Enders-Weller-Robbins technique to produce large quantities of poliovirus, and then developed a polio vaccine in 1952. Upon the 1954 polio vaccine field trial, whose success Salk announced on the radio, Salk became a public hero but failed to credit the many other researchers that his effort rode upon, and was somewhat shunned by America's scientific establishment.
In 1954, Enders and Thomas C. Peebles isolated measlesvirus from an 11-year-old boy, David Edmonston. Disappointed by polio vaccine's development and involvement in some cases of polio and death—what Enders attributed to Salk's technique—Enders began development of measles vaccine. In October 1960, an Enders team began trials on 1,500 intellectually disabled children in New York City and on 4,000 children in Nigeria. Refusing credit for merely himself when The New York Times announced the measles vaccine effective on September 17, 1961, Enders wrote to the newspaper to acknowledge the work of various colleagues and the collaborative nature of the research. In 1963, a deactivated measles vaccine and an attenuated measles vaccine were introduced by Pfizer and Merck & Co., respectively.
He continued to work in virology research till the late 1970s and retired from the laboratory at the age of 80.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1889 2026-03-08 00:07:46
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2452) Frederick Chapman Robbins
Gist:
Work
Many infectious diseases are caused by viruses—very small biological particles. A virus lacks metabolism of its own and cannot multiply without infecting a living cell. For a long time the prevailing opinion was that viruses could not be cultured in a laboratory. However, in 1949 Thomas Weller, John Enders, and Frederick Robbins succeeded in culturing the virus that causes polio in human muscle and tissue in a laboratory setting. This became an important step on the road toward a vaccine against polio.
Summary
Frederick Chapman Robbins (born August 25, 1916, Auburn, Alabama, U.S.—died August 4, 2003, Cleveland, Ohio) was an American pediatrician and virologist who received (with John Enders and Thomas Weller) the 1954 Nobel Prize for Physiology or Medicine for successfully cultivating poliomyelitis virus in tissue cultures. This accomplishment made possible the production of polio vaccines, the development of sophisticated diagnostic methods, and the isolation of new viruses.
A graduate of Harvard University Medical School (1940), Robbins served in the United States, Italy, and North Africa during World War II (1942–46) as chief of the U.S. Army’s 15th medical general laboratory virus and rickettsia section, where he investigated epidemics of infectious hepatitis, typhus, and Q fever.
After joining Enders and Weller at the Children’s Hospital in Boston in 1948, Robbins helped solve the difficult problem of propagating viruses—then known to grow only in living organisms—in laboratory suspensions of actively metabolizing cells in nutrient solutions. At that time it was believed that the virus responsible for poliomyelitis grew and multiplied only in mammalian nerve tissue, which is highly difficult to maintain outside the living animal. By 1952 Robbins and his colleagues had succeeded in cultivating the virus in mixtures of human embryonic skin and muscle tissue suspended in cell cultures, dramatically demonstrating that the polio virus subsists in extraneural tissue, only later attacking the lower part of the brain and sections of the spinal cord.
Robbins served as director of the department of pediatrics and contagious diseases at the Cleveland Metropolitan General Hospital (1952–66) and as professor of pediatrics (1952–80) and dean (1966–80) at the Case Western Reserve University School of Medicine, Cleveland, Ohio. He later served as president of the Institute of Medicine of the National Academy of Sciences (1980–85).
Details
Frederick Chapman Robbins (August 25, 1916 – August 4, 2003) was an American pediatrician and virologist. He was born in Auburn, Alabama, and grew up in Columbia, Missouri, attending David H. Hickman High School.
He received the Nobel Prize in Physiology or Medicine in 1954 along with John Franklin Enders and Thomas Huckle Weller, making Robbins the only Nobel laureate born in Alabama. The award was for breakthrough work in isolating and growing the poliovirus in tissue culture, paving the way for vaccines developed by Jonas Salk and Albert Sabin. He attended the University of Missouri and Harvard University.
In 1952, he was appointed professor of pediatrics at Case Western Reserve University. Robbins was elected a fellow of the American Academy of Arts and Sciences in 1962. From 1966 to 1980, Robbins was dean of the School of Medicine at Case Western. He was elected to the American Philosophical Society in 1972. In 1980, he assumed the presidency of the National Academy of Sciences' Institute of Medicine. He had been a member of the National Academy of Sciences since 1972. Five years later, in 1985, Robbins returned to Case Western Reserve as dean emeritus and distinguished university professor emeritus. He continued to be a fixture at the medical school until his death in 2003. The medical school's Frederick C. Robbins Society is named in his honor. His wife, Alice N. Robbins, died in 2016. She was the daughter of Nobel laureate John Howard Northrop.
Robbins received the Benjamin Franklin Medal for Distinguished Achievement in the Sciences of the American Philosophical Society in 1999.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1890 2026-03-09 00:03:02
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2453) Thomas Huckle Weller
Gist:
Work
Many infectious diseases are caused by viruses—very small biological particles. A virus lacks metabolism of its own and cannot multiply without infecting a living cell. For a long time the prevailing opinion was that viruses could not be cultured in a laboratory. However, in 1949 Frederick Robbins, John Enders, and Thomas Weller succeeded in culturing the virus that causes polio in human muscle and tissue in a laboratory setting. This became an important step on the road toward a vaccine against polio.
Summary
Thomas H. Weller (born June 15, 1915, Ann Arbor, Mich., U.S.—died Aug. 23, 2008, Needham, Mass.) was an American physician and virologist who was the corecipient (with John Enders and Frederick Robbins) of the Nobel Prize for Physiology or Medicine in 1954 for the successful cultivation of poliomyelitis virus in tissue cultures. This made it possible to study the virus “in the test tube”—a procedure that led to the development of polio vaccines.
After his education at the University of Michigan at Ann Arbor (A.B., 1936; M.S., 1937) and Harvard University (M.D., 1940), Weller became a teaching fellow at the Harvard Medical School (1940–42) and served in the U.S. Army Medical Corps during World War II. He was appointed assistant director of Enders’ infectious diseases laboratory at the Children’s Medical Center, Boston (1949–55), and, working with Enders and Robbins, soon achieved the propagation of poliomyelitis virus in laboratory suspensions of human embryonic skin and muscle tissue. He was also the first (with the American physician Franklin Neva) to achieve the laboratory propagation of rubella (German measles) virus and to isolate chicken pox virus from human cell cultures. Weller became professor of tropical public health at Harvard University in 1954 and from 1966 to 1981 served also as director of the Center for the Prevention of Infectious Diseases at the Harvard University School of Public Health. Weller’s autobiography, Growing Pathogens in Tissue Cultures: Fifty Years in Academic Tropical Medicine, Pediatrics, and Virology, was published in 2004.
Details
Thomas Huckle Weller (June 15, 1915 – August 23, 2008) was an American virologist. He, John Franklin Enders and Frederick Chapman Robbins were awarded a Nobel Prize in Physiology or Medicine in 1954 for showing how to cultivate poliomyelitis viruses in a test tube, using a combination of human embryonic skin and muscle tissue.
Biography
Weller was born and grew up in Ann Arbor, Michigan, where he graduated from Ann Arbor High School. He then went to the University of Michigan, where his father Carl Vernon Weller was a professor in the Department of Pathology. At Michigan, he studied medical zoology and received a B.S. and an M.S., with his masters thesis on fish parasites.
In 1936, Weller entered Harvard Medical School, and in 1939 began working under John Franklin Enders, with whom he would later (along with Frederick Chapman Robbins) share the Nobel Prize. It was Enders who got Weller involved in researching viruses and tissue-culture techniques for determining infectious disease causes. Weller received his MD in 1940, and went to work at Children's Hospital in Boston.
In 1942, during World War II, he entered the Army Medical Corps and was stationed at the Antilles Medical Laboratory in Puerto Rico, earning the rank of Major and heading the facility's Departments of Bacteriology, Virology and Parasitology. After the War, he returned to Children's Hospital in Boston, and it was there in 1947, that he rejoined Enders in the newly created Research Division of Infectious Diseases. After several leading positions, in July 1954, he was appointed head of the Tropical Public Health Department at the Harvard School of Public Health. Weller also served from 1953 to 1959 as director of the Commission on Parasitic Diseases of the American Armed Forces Epidemiological Board.
In addition to his work on polio, Weller also contributed to treating schistosomiasis, and Coxsackie viruses. He was also the first to isolate the virus responsible for chickenpox.
Awards
In 1954, Weller was awarded the George Ledlie prize in recognition of his research on rubella, polio and cytomegalovirus(CMV) viruses.
He was awarded the Nobel Prize in Physiology or Medicine in 1954 for his research on polio.
In 1996 he was awarded the Walter Reed Medal from the American Society of Tropical Medicine and Hygiene.
Personal life
In 1945, Weller married Kathleen Fahey, who died in 2011 aged 95. They had two sons and two daughters.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1891 2026-03-10 00:04:31
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2454) Frits Zernike
Gist:
Work
When light passes through a transparent object, there is a change in the phase of the light waves, the position of the wave crests’ in relation to one another. Our eye does not perceive this, but in the beginning of the 1930s, Frits Zernike developed a way to make it visible. A light beam is passed through an object while a reference beam goes by it. When the beams are brought together, they are strengthened or canceled out because of phase displacement, and the object is outlined more clearly in contrast to its surroundings. The phase contrast microscope became particularly important in the study of living cells.
Summary
Frits Zernike (born July 16, 1888, Amsterdam, Neth.—died March 10, 1966, Groningen) was a Dutch physicist, winner of the Nobel Prize for Physics in 1953 for his invention of the phase-contrast microscope, an instrument that permits the study of internal cell structure without the need to stain and thus kill the cells.
Zernike obtained a doctorate from the University of Amsterdam in 1915. He became an assistant at the State University of Groningen in 1913 and served as a full professor there from 1920 to 1958. His earliest work in optics was concerned with astronomical telescopes. While studying the flaws that occur in some diffraction gratings because of the imperfect spacing of engraved lines, he discovered the phase-contrast principle. He noted that he could distinguish the light rays that passed through different transparent materials. He built a microscope using that principle in 1938. In 1952 Zernike was awarded the Rumford Medal of the Royal Society of London.
Details
Frits Zernike (16 July 1888 – 10 March 1966) was a Dutch physicist who received the Nobel Prize in Physics in 1953 for his invention of the phase-contrast microscope.
Early life and education
Frederick Zernike was born on 16 July 1888 in Amsterdam, Netherlands to Carl Friedrich August Zernike and Antje Dieperink. Both parents were teachers of mathematics, and he especially shared his father's passion for physics. In 1905 he enrolled at the University of Amsterdam, studying chemistry (his major), mathematics and physics.
Academic career
In 1912, he was awarded a prize for his work on opalescence in gases. In 1913, he became assistant to Jacobus Kapteyn at the astronomical laboratory of Groningen University. In 1914, Zernike and Leonard Ornstein were jointly responsible for the derivation of the Ornstein–Zernike equation in critical-point theory. In 1915, he became lector in theoretical mechanics and mathematical physics at the same university and in 1920 he was promoted to professor of mathematical physics.
In 1930, Zernike was conducting research into spectral lines when he discovered that the so-called ghost lines that occur to the left and right of each primary line in spectra created by means of a diffraction grating, have their phase shifted from that of the primary line by 90 degrees. It was at a Physical and Medical Congress in Wageningen in 1933, that Zernike first described his phase contrast technique in microscopy. He extended his method to test the figure of concave mirrors. His discovery lay at the base of the first phase contrast microscope, built during World War II.
He also made another contribution in the field of optics, relating to the efficient description of the imaging defects or aberrations of optical imaging systems like microscopes and telescopes. The representation of aberrations was originally based on the theory developed by Ludwig Seidel in the middle of the nineteenth century. Seidel's representation was based on power series expansions and did not allow a clear separation between various types and orders of aberrations. Zernike's orthogonal circle polynomials provided a solution to the long-standing problem of the optimum 'balancing' of the various aberrations of an optical instrument. Since the 1960s, Zernike's circle polynomials are widely used in optical design, optical metrology and image analysis.
Zernike's work helped awaken interest in coherence theory, the study of partially coherent light sources. In 1938 he published a simpler derivation of Van Cittert's 1934 theorem on the coherence of radiation from distant sources, now known as the Van Cittert–Zernike theorem.
Death
He died in hospital in Amersfoort in 1966 after suffering illness the last years of his life. His granddaughter is the journalist Kate Zernike.
Honours and awards
In 1946, Zernike became member of the Royal Netherlands Academy of Arts and Sciences.
In 1953, Zernike won the Nobel Prize in Physics, for his invention of the phase-contrast microscope, an instrument that permits the study of internal cell structure without the need to stain and thus kill the cells.
In 1954, Zernike became an Honorary Member of The Optical Society (OSA). Zernike was elected a Foreign Member of the Royal Society.
The university complex (Zernike Campus) to the north of the city of Groningen is named after him, as is the crater Zernike on the Moon and the minor planet 11779 Zernike.
Zernike's great-nephew Gerard 't Hooft won the Nobel Prize in Physics in 1999.
The Oz Enterprise, a Linux distribution, was named after Leonard Ornstein and Frederik Zernike.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1892 2026-03-11 00:04:29
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2455) Hermann Staudinger
Gist:
Work
The world around us is made of atoms combined to form molecules. In the early 1900s chemists debated how large these molecules could become. In the early 1920s Herman Staudinger claimed they could be very large; tens or even hundreds of thousands of atoms in size. He showed how small molecules can join to form long chains and so become very large molecules—polymers. The result was the basis for the development of synthetic materials like plastics.
Summary
Hermann Staudinger (born March 23, 1881, Worms, Germany—died September 8, 1965, Freiburg im Breisgau, West Germany [now Germany]) was a German chemist who won the 1953 Nobel Prize for Chemistry for demonstrating that polymers are long-chain molecules. His work laid the foundation for the great expansion of the plastics industry later in the 20th century.
Staudinger studied chemistry at the universities of Darmstadt and Munich, and he received a Ph.D. from the University of Halle in 1903. He held academic posts at the universities of Strassburg (now Strasbourg) and Karlsruhe before joining the faculty at the Swiss Federal Institute of Technology in Zürich in 1912. He left the institute in 1926 to become a lecturer at the Albert Ludwig University of Freiburg im Breisgau, where in 1940 an Institute for Macromolecular Chemistry was established under his directorship. Staudinger’s wife, the Latvian plant physiologist Magda Woit, was his coworker and coauthor. He retired in 1951.
Staudinger’s first discovery was that of the highly reactive organic compounds known as ketenes. His work on polymers began with research he conducted for the German chemical firm BASF on the synthesis of isoprene (1910), the monomer of which natural rubber is composed. The prevalent belief at the time was that rubber and other polymers are composed of small molecules that are held together by “secondary” valences or other forces. In 1922 Staudinger and J. Fritschi proposed that polymers are actually giant molecules (macromolecules) that are held together by normal covalent bonds, a concept that met with resistance from many authorities. Throughout the 1920s, the researches of Staudinger and others showed that small molecules form long, chainlike structures (polymers) by chemical interaction and not simply by physical aggregation. Staudinger showed that such linear molecules could be synthesized by a variety of processes and that they could maintain their identity even when subject to chemical modification.
Staudinger’s pioneering work provided the theoretical basis for polymer chemistry and greatly contributed to the development of modern plastics. His researches on polymers eventually contributed to the development of molecular biology, which seeks to understand the structure of proteins and other macromolecules found in living organisms. Staudinger wrote numerous papers and books, including Arbeitserinnerungen (1961; “Working Memories”). Two of his students, Leopold Ružička and Tadeus Reichstein, also won Nobel Prizes.
Details
Hermann Staudinger (23 March 1881 – 8 September 1965) was a German organic chemist who demonstrated the existence of macromolecules, which he characterized as polymers. For this work he received the 1953 Nobel Prize in Chemistry.
He is also known for his discovery of ketenes and of the Staudinger reaction. Staudinger, together with Leopold Ružička, also elucidated the molecular structures of pyrethrin I and II in the 1920s, enabling the development of pyrethroid insecticides in the 1960s and 1970s.
Early work
Staudinger was born in 1881 in Worms. Staudinger, who initially wanted to become a botanist, studied chemistry at the University of Halle, at the TH Darmstadt and at the LMU Munich. He received his "Verbandsexamen" (comparable to Master's degree) from TH Darmstadt. After receiving his Ph.D. from the University of Halle in 1903, Staudinger qualified as an academic lecturer at the University of Strasbourg in 1907. He was supported in his work by his new wife Dora Staudinger who wrote up his lectures.
It was here that he discovered the ketenes, a family of molecules characterized by the general form depicted in Figure 1. Ketenes would prove a synthetically important intermediate for the production of yet-to-be-discovered antibiotics such as penicillin and amoxicillin.
In 1907, Staudinger began an assistant professorship at the Technical University of Karlsruhe. Here, he successfully isolated a number of useful organic compounds (including a synthetic coffee flavoring) as more completely reviewed by Rolf Mülhaupt. Here too he guided future Nobel laureates Leopold Ružička (1910) and Tadeusz Reichstein to their doctorates.
The Staudinger reaction
In 1912, Staudinger took on a new position at the Swiss Federal Institute of Technology in Zurich, Switzerland. One of his earliest discoveries came in 1919, when he and colleague Meyer reported that organic azides react with triphenylphosphine to form an iminophosphorane. This reaction, commonly referred to as the Staudinger reaction, typically produces a high yield of the iminophosphorane.
World War I
While in autumn 1914 German professors joined the widespread public support of the war, Staudinger refused to sign Manifesto of the Ninety-Three and joined the few exceptions like Max Born, Otto Buek and Albert Einstein in condemning it. In 1917 he authored an essay predicting the defeat of Germany due to industrial superiority of the Entente and called for a peaceful settlement as soon as possible, and after the entrance of the US he repeated the call in a long letter to the German military leadership. Fritz Haber attacked him for his essay, accusing him of harming Germany, and Staudinger in turn criticized Haber for his role in the German chemical weapons program.
Polymer chemistry
While at Karlsruhe and later, Zurich, Staudinger began research in the chemistry of rubber, for which very high molecular weights had been measured by the physical methods of Raoult and van 't Hoff. Contrary to prevailing ideas, Staudinger proposed in a landmark paper published in 1920 that rubber and other polymers such as starch, cellulose and proteins are long chains of short repeating molecular units linked by covalent bonds. In other words, polymers are like chains of paper clips, made up of small constituent parts linked from end to end.
At the time, leading organic chemists such as Emil Fischer and Heinrich Wieland believed that the measured high molecular weights were only apparent values caused by the aggregation of small molecules into colloids. At first, the majority of Staudinger’s colleagues refused to accept the possibility that small molecules could link together covalently to form high-molecular weight compounds. As Mülhaupt aptly notes, this is due in part to the fact that molecular structure and bonding theory were not fully understood in the early 20th century.
In 1926, he was appointed lecturer of chemistry at the University of Freiburg at Freiburg im Breisgau (Germany), where he spent the rest of his career. Further evidence to support his polymer hypothesis emerged in the 1930s. High molecular weights of polymers were confirmed by membrane osmometry, and also by Staudinger’s measurements of viscosity in solution. The X-ray diffraction studies of polymers by Herman Mark provided direct evidence for long chains of repeating molecular units. And the synthetic work led by Carothers demonstrated that polymers such as nylon and polyester could be prepared by well-understood organic reactions. His theory opened up the subject to further development, and helped place polymer science on a sound basis.
Private life
He married in 1906 to Dora Förster and they remained together until their divorce in 1926. They had four children including Eva Lezzi (1907-1993) and Klar (Klara) Kaufmann who were active in resisting the rise of fascism. Dora married again and became a leading peace activist.
In 1927, he married the Latvian botanist, Magda Voita (German: Magda Woit), who was a collaborator with him until his death and whose contributions he acknowledged in his Nobel Prize acceptance.
In 1935 Staudinger became a Patron Member of the SS.
Legacy
Staudinger's groundbreaking elucidation of the nature of the high-molecular weight compounds he termed Makromoleküle paved the way for the birth of the field of polymer chemistry. Staudinger himself saw the potential for this science long before it was fully realized. "It is not improbable," Staudinger commented in 1936, "that sooner or later a way will be discovered to prepare artificial fibers from synthetic high-molecular products, because the strength and elasticity of natural fibers depend exclusively on their macro-molecular structure – i.e., on their long thread-shaped molecules." Staudinger founded the first polymer chemistry journal in 1940, and in 1953 received the Nobel Prize in Chemistry for "his discoveries in the field of macromolecular chemistry." In 1999, the American Chemical Society and the German Chemical Society designated Staudinger's work as an International Historic Chemical Landmark. His pioneering research has afforded the world myriad plastics, textiles, and other polymeric materials which make consumer products more affordable, attractive and enjoyable, while helping engineers develop lighter and more durable structures. The German Chemical Society started the Hermann Staudinger Prize in 1971 to recognize fundamental contributions in polymer science.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1893 2026-03-12 00:05:57
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2456) Hans Krebs
Gist:
Work
Nutrients are broken down in our cells to release energy for the construction of cells. After Albert Szent-Györgyi identified several important reactions in these metabolic processes, in 1937 Hans Krebs was able to present a complete picture of an important part of metabolism—the citric acid cycle. In this process, which is cyclical and has several steps, nutrients are converted to other molecules with a large amount of chemical energy. The latter are ultimately converted into adenosine triphosphate (ATP), which provides chemical energy to facilitate other biochemical processes in the cell.
Summary
Sir Hans Adolf Krebs (born Aug. 25, 1900, Hildesheim, Ger.—died Nov. 22, 1981, Oxford, Eng.) was a German-born British biochemist who received (with Fritz Lipmann) the 1953 Nobel Prize for Physiology or Medicine for the discovery in living organisms of the series of chemical reactions known as the tricarboxylic acid cycle (also called the citric acid cycle, or Krebs cycle). These reactions involve the conversion—in the presence of oxygen—of substances that are formed by the breakdown of sugars, fats, and protein components to carbon dioxide, water, and energy-rich compounds.
At the University of Freiburg (1932), Krebs discovered (with the German biochemist Kurt Henseleit) a series of chemical reactions (now known as the urea cycle) by which ammonia is converted to urea in mammalian tissue; the urea, far less toxic than ammonia, is subsequently excreted in the urine of most mammals. This cycle also serves as a major source of the amino acid arginine.
The son of a Jewish physician, Krebs was forced in 1933 to leave Nazi Germany for England, where he continued his research at the University of Cambridge (1933–35). At Sheffield University, Yorkshire (1935–54), Krebs measured the amounts of certain four-carbon and six-carbon acids generated in pigeon liver and breast muscle when sugars are oxidized completely to yield carbon dioxide, water, and energy.
In 1937 Krebs demonstrated the existence of a cycle of chemical reactions that combines the end-product of sugar breakdown, later shown to be an “activated” form of the two-carbon acetic acid, with the four-carbon oxaloacetic acid to form citric acid. The cycle regenerates oxaloacetic acid through a series of intermediate compounds while liberating carbon dioxide and electrons that are immediately utilized to form high-energy phosphate bonds in the form of adenosine triphosphate (ATP; the chemical-energy reservoir of the cell). The discovery of the tricarboxylic acid cycle, which is central to nearly all metabolic reactions and the source of two-thirds of the food-derived energy in higher organisms, was of vital importance to a basic understanding of cell metabolism and molecular biology.
Krebs served on the faculty of the University of Oxford from 1954 to 1967. He wrote (with the British biochemist Hans Kornberg) Energy Transformations in Living Matter (1957) and also coauthored (with Anne Martin) Reminiscences and Reflections (1981). He was knighted in 1958, and the Royal Society awarded him its Copley Medal in 1961.
Details
Sir Hans Adolf Krebs, (25 August 1900 – 22 November 1981) was a German-British biologist, physician and biochemist. He was a pioneer scientist in the study of cellular respiration, a biochemical process in living cells that extracts energy from food and oxygen and makes it available to drive the processes of life. He is best known for his discoveries of two important sequences of chemical reactions that take place in the cells of nearly all organisms, including humans, other than anaerobic microorganisms, namely the citric acid cycle and the urea cycle. The former, often eponymously known as the "Krebs cycle", is the sequence of metabolic reactions that allows cells of oxygen-respiring organisms to obtain far more ATP from the food they consume than anaerobic processes such as glycolysis can supply; and its discovery earned Krebs a Nobel Prize in Physiology or Medicine in 1953. With Hans Kornberg, he also discovered the glyoxylate cycle,[8] a slight variation of the citric acid cycle found in plants, bacteria, protists, and fungi.
Krebs died in 1981 in Oxford, where he had spent 13 years of his career from 1954 until his retirement in 1967 at the University of Oxford.
Biography:
Early life and education
Krebs was born in Hildesheim, Germany, to Georg Krebs, an ear, nose, and throat surgeon, and Alma Krebs (née Davidson). He was of Jewish ancestry and was the middle of three children. He had an elder sister, Elisabeth, and a younger brother, Wolfgang.
Krebs attended school at the Gymnasium Andreanum in his home town. Near the end of World War I, in September 1918, six months short of completing his secondary school education, he was conscripted into the Imperial German Army. He was allowed to take an emergency examination for his high school diploma, which he passed with such a high score that he suspected the examiners of being "unduly lenient and sympathetic". With the end of the war two months later, his conscription ended.
Krebs decided to follow his father's profession and entered the University of Göttingen in December 1918 to study medicine. In 1919, he transferred to the University of Freiburg. In 1923, he published his first scientific paper on a tissue staining technique. He did this work under the guidance of Wilhelm von Mollendorf starting it in 1920. He completed his medical course in December 1923. To obtain a Doctor of Medicine degree, and a medical license, he spent one year at the Third Medical Clinic in the University of Berlin. By then he had turned his professional goal from becoming a practising physician to becoming a medical researcher, particularly in biochemistry. In 1924, he studied at the Department of Chemistry at the Pathological Institute of the Charité Hospital, in Berlin, for training in chemistry and biochemistry. He earned his MD degree in 1925 from the University of Hamburg.
Career
In 1926, Krebs joined Otto Heinrich Warburg as a research assistant at the Kaiser Wilhelm Institute for Biology in Dahlem, Berlin. He was paid 4800 marks per year. After four years in 1930, with 16 publications to his credit, his mentor Warburg urged him to move on and he took up the position of Assistant in the Department of Medicine at the Municipal Hospital in Altona (now part of Hamburg). The next year he moved to the Medical Clinic of the University of Freiburg. At Freiburg, he was in charge of about 40 patients, and was at liberty to do his own research. Before a year was over at Freiburg, he, with research student Kurt Henseleit, published their discovery of the ornithine cycle of urea synthesis, which is the metabolic pathway for urea formation. It is now known as the urea cycle, and is sometimes also referred to as the Krebs–Henseleit cycle. Together they also developed a complex aqueous solution (a buffer), or perfusion ex vivo, for studying blood flow in arteries, which is now called the Krebs–Henseleit buffer. In 1932, he published the basic chemical reactions of the urea cycle, which established his scientific reputation.
Krebs's life as a respected German scientist came to an abrupt halt in 1933 because of his Jewish ancestry. With the rise of Hitler's Nazi Party to power, Germany decreed the Law for the Restoration of the Professional Civil Service, which decreed the removal of all non-Germans, and anti-Nazis, from professional occupations. Krebs received his official dismissal from his job in April 1933, and his service was terminated on 1 July 1933. An admirer, Sir Frederick Gowland Hopkins at the University of Cambridge, immediately came to his rescue, and persuaded the university to recruit Krebs to work with him in the Department of Biochemistry. By July 1933, he was settled in Cambridge with financial support from the Rockefeller Foundation.
Although Germany restricted him to bringing only his personal belongings, he was fortunate that the government agents allowed him to take his equipment and research samples to England. They proved to be pivotal to his later discoveries, especially the manometer developed by Warburg specifically for the measurement of oxygen consumption in thin slices of tissues; it was the basis for his research.
He was appointed as Demonstrator in biochemistry in 1934, and in 1935 the University of Sheffield offered him a post of Lecturer in Pharmacology, with a more spacious laboratory and double the salary. He worked there for 19 years. The University of Sheffield opened a Department of Biochemistry, now Department of Molecular Biology and Biotechnology, in 1938 and Krebs became its first Head, and eventually a Professor in 1945. During his time it became one of the leading departments of biochemistry in the world. Krebs took over the running of the Sorby Research Institute in 1943. In 1944, the British Medical Research Council established the MRC Unit for Cell Metabolism Research at Sheffield, and Krebs was appointed the Director. With this, his laboratory became so large that the locals jokingly nicknamed it "Krebs's Empire".
He moved with his MRC unit to the University of Oxford in 1954 as Whitley Professor of Biochemistry, the post he held until his retirement in 1967. The editorial board of Biochemical Journal extended their good wishes on his retirement, but in return he promised to keep them busy, by producing scientific papers. He continued his research, and took his MRC unit to the Nuffield Department of Clinical Medicine at the Radcliffe Infirmary, Oxford. From there he published over 100 research papers.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1894 2026-03-13 00:06:15
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2457) Fritz Albert Lipmann
Gist:
Work
In 1937 Hans Krebs presented a complete picture of an important part of metabolism—the citric acid cycle. In this process, which has several steps, nutrients are converted to other molecules with a large amount of chemical energy. An important piece of the process was still missing—a substance that along with a protein forms an enzyme that facilitates an important step. In 1946 the substance was discovered by Fritz Lipmann, who described its role plays and gave it the name coenzyme A.
Summary
Fritz Albert Lipmann (born June 12, 1899, Königsberg, Ger. [now Kaliningrad, Russia]—died July 24, 1986, Poughkeepsie, N.Y., U.S.) was a German-born American biochemist, who received (with Sir Hans Krebs) the 1953 Nobel Prize for Physiology or Medicine for the discovery of coenzyme A, an important catalytic substance involved in the cellular conversion of food into energy.
Lipmann earned an M.D. degree (1924) and a Ph.D. degree (1927) from the University of Berlin. He conducted research in the laboratory of the biochemist Otto Meyerhof at the University of Heidelberg (1927–30) and then did research at the Biological Institute of the Carlsberg Foundation (Carlsbergfondets Biologiske Institut), Copenhagen (1932–39), and at the Cornell Medical School, New York City (1939–41).
At Massachusetts General Hospital, Boston (1941–57), where he directed the biochemistry research department, and as professor of biological chemistry at the Harvard Medical School (1949–57), Lipmann found a catalytically active, heat-stable factor in pigeon liver extracts. He subsequently isolated (1947), named, and determined the molecular structure (1953) of this factor, coenzyme A (or CoA), which is now known to be bound to acetic acid as the end product of sugar and fat breakdown in the absence of oxygen. Coenzyme A is one of the most important substances involved in cellular metabolism; it helps in the conversion of amino acids, steroids, fatty acids, and hemoglobins into energy.
Lipmann taught or conducted research at the Rockefeller Institute, now Rockefeller University, New York City, from 1957 until his death.
Details
Fritz Albert Lipmann (June 12, 1899 – July 24, 1986) was a German-American biochemist and a co-discoverer in 1945 of coenzyme A. For this, together with other research on coenzyme A, he was awarded the Nobel Prize in Physiology or Medicine in 1953 (shared with Hans Adolf Krebs).
Early life and education
Lipmann was born in Königsberg, Germany, to a Jewish family. His parents were Gertrud (Lachmanski) and Leopold Lipmann, an attorney.
Lipmann studied medicine at the University of Königsberg, Berlin, and Munich, graduating in Berlin in 1924. He returned to Königsberg to study chemistry under Professor Hans Meerwein. In 1926 he joined Otto Meyerhof at the Kaiser Wilhelm Institute for Biology, Dahlem, Berlin, for his doctoral thesis. After that he followed Meyerhof to Heidelberg to the Kaiser Wilhelm Institute for Medical Research.
Career
From 1939 on, Lipmann lived and worked in the United States. He was a Research Associate in the Department of Biochemistry, Cornell University Medical College, New York from 1939 to 1941. He joined the research staff of the Massachusetts General Hospital in Boston in 1941, first as a Research Associate in the Department of Surgery, then heading his own group in the Biochemical Research Laboratory of the hospital. From 1949 to 1957 he was professor of biological chemistry at Harvard Medical School. From 1957 onwards, he taught and conducted research at Rockefeller University, New York City.
In 1953, Lipmann received one half of the Nobel Prize in Physiology and Medicine "for his discovery of co-enzyme A and its importance for intermediary metabolism." The other half of the award was won by Hans Adolf Krebs. Lipmann was awarded the National Medal of Science in 1966. He would try to dive further into his discovery by finding a variant of co-enzyme A, now known as Pantethine. He was an elected member of the American Academy of Arts and Sciences, the United States National Academy of Sciences, and the American Philosophical Society.
Lipmann introduced the specific squiggle designation (~) to indicate high energy-rich phosphate in energy-rich biomolecules like ATP in his essay "Metabolic Generation and Utilization of Phosphate Bond Energy." Of his work, he said "that in the field of biosynthesis we have a rare example of progress leading to simplification."
Personal life
In 1931, Lipmann married Elfreda M. Hall. They had one son.[2] Lipmann died in New York in 1986.The photo shows Mary Soames, not Elfreda Hall Lipmann[10] His widow died in 2008 at the age of 101.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1895 2026-03-14 00:04:00
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2458) Felix Bloch
Gist:
Life
Felix Bloch was born in Zurich, Switzerland, the son of a merchant, and studied at ETH and elsewhere. When the Nazis took power in 1933, he left Europe to work at Stanford University. After becoming an American citizen, he worked on atomic energy in Los Alamos during World War II and later on radar at Harvard University. Immediately after the war, he did his Nobel Prize-awarded work at Stanford. He became the first head of CERN outside Geneva in 1954-1955. Bloch was married and had four children.
Work
Protons and neutrons in nuclei act like small, rotating magnets. Atoms and molecules therefore align in a magnetic field. Radio waves can disturb their direction of rotation, but only in certain stages, in accordance with quantum mechanics. When the atoms return to their original positions, they emit electromagnetic radio waves with frequencies characteristic of different elements and isotopes. In 1946, Felix Bloch and Edward Purcell developed methods for precise measurement, making it possible to study different materials’ compositions.
Summary
Felix Bloch (born Oct. 23, 1905, Zürich, Switz.—died Sept. 10, 1983, Zürich) was a Swiss-born American physicist who shared (with E.M. Purcell) the Nobel Prize for Physics in 1952 for developing the nuclear magnetic resonance method of measuring the magnetic field of atomic nuclei.
Bloch’s doctoral dissertation (University of Leipzig, 1928) promulgated a quantum theory of solids that provided the basis for understanding electrical conduction. Bloch taught at the University of Leipzig until 1933; when Adolf Hitler came to power he emigrated to the United States and was naturalized in 1939. After joining the faculty of Stanford University, Palo Alto, Calif., in 1934, he proposed a method for splitting a beam of neutrons into two components that corresponded to the two possible orientations of a neutron in a magnetic field. In 1939, using this method, he and Luis Alvarez (winner of the Nobel Prize for Physics in 1968) measured the magnetic moment of the neutron (a property of its magnetic field). Bloch worked on atomic energy at Los Alamos, N.M., and radar countermeasures at Harvard University during World War II.
Bloch returned to Stanford in 1945 to develop, with physicists W.W. Hansen and M.E. Packard, the principle of nuclear magnetic resonance, which helped establish the relationship between nuclear magnetic fields and the crystalline and magnetic properties of various materials. It later became useful in determining the composition and structure of molecules. Nuclear magnetic resonance techniques have become increasingly important in diagnostic medicine.
Bloch was the first director general of the European Organization for Nuclear Research (1954–55; CERN).
Details
Felix Bloch (23 October 1905 – 10 September 1983) was a Swiss-American theoretical physicist who shared the 1952 Nobel Prize in Physics with Edward Mills Purcell "for their development of new methods for nuclear magnetic precision measurements and discoveries in connection therewith".
He was the first Stanford University Nobel laureate.
Bloch made fundamental theoretical contributions to the understanding of ferromagnetism and electron behavior in crystal lattices. He is also considered one of the developers of nuclear magnetic resonance.
Education
Bloch was born on 23 October 1905 in Zurich, Switzerland, to Jewish parents, Gustav Bloch and Agnes Mayer. Gustav was financially unable to attend university and worked as a wholesale grain dealer in Zurich. Gustav moved to Zurich from Moravia in 1890 to become a Swiss citizen. Their first child was a girl born in 1902, while Felix was born three years later.
Bloch entered public elementary school at the age of six and is said to have been teased, in part because he "spoke Swiss German with a somewhat different accent than most members of the class". He received support from his older sister during much of this time, but she died at the age of 12, devastating Felix, who is said to have lived a "depressed and isolated life" in the following years. Bloch learned to play the piano by the age of 8 and was drawn to arithmetic for its "clarity and beauty". Bloch graduated from elementary school at twelve and enrolled in the Cantonal Gymnasium in Zurich for secondary school in 1918. He was placed on a six-year curriculum here to prepare him for university. He continued his curriculum through 1924, even through his study of engineering and physics in other schools, though it was limited to mathematics and languages after the first three years.
After these first three years at the Gymnasium, at the age of 15, Bloch began to study at the ETH Zurich. Although he initially studied engineering, he soon changed to physics. During this time, he attended lectures and seminars given by Peter Debye and Hermann Weyl at the ETH Zurich and Erwin Schrödinger at the neighboring University of Zurich. A fellow student in these seminars was John von Neumann.
Bloch graduated in 1927, and was encouraged by Debye to go to the University of Leipzig to study under Werner Heisenberg. Bloch became Heisenberg's first graduate student, and gained his doctorate in 1928. His doctoral thesis established the quantum theory of solids, using waves to describe electrons in periodic lattices.
Career and research
Bloch remained in European academia, working on superconductivity with Wolfgang Pauli in Zurich; with Hans Kramers and Adriaan Fokker in the Netherlands; with Heisenberg on ferromagnetism, where he developed a description of boundaries between magnetic domains, now known as Bloch walls, and theoretically proposed a concept of spin waves, excitations of magnetic structure; with Niels Bohr in Copenhagen, where he worked on a theoretical description of the stopping of charged particles traveling through matter; and with Enrico Fermi in Rome.
In 1932, Bloch returned to Leipzig to assume a position as Privatdozent (lecturer). In 1933, immediately after Adolf Hitler came to power, Bloch left Germany out of fear of anti-Jewish persecution, returning to Zurich before traveling to Paris to lecture at the Institut Henri Poincaré.
In 1934, the chairman of Stanford Physics invited Bloch to join the faculty. Bloch accepted the offer and emigrated to the United States. In the fall of 1938, Bloch began working with the 37 inch cyclotron at the University of California, Berkeley, to determine the magnetic moment of the neutron. Bloch went on to become the first professor of theoretical physics at Stanford. In 1939, he became a naturalized citizen of the United States.
During World War II, Bloch briefly worked on the atomic bomb project at Los Alamos. Disliking the military atmosphere of the laboratory and uninterested in the theoretical work there, Bloch left to join the radar project at Harvard University.
After the war, he concentrated on investigations into nuclear induction and nuclear magnetic resonance, which are the underlying principles of MRI. In 1946, he proposed the Bloch equations, which determine the time evolution of nuclear magnetization. He was elected to the National Academy of Sciences in 1948. Along with Edward Purcell, Bloch was awarded the Nobel Prize in Physics in 1952 for his work on nuclear magnetic induction.
When CERN was being set up in the early 1950s, its founders were searching for someone of stature and international prestige to head the fledgling international laboratory, and in 1954 Professor Bloch became CERN's first director-general, at the time when construction was getting under way on the present Meyrin site and plans for the first machines were being drawn up. After leaving CERN, he returned to Stanford University, where he in 1961 was made Max Stein Professor of Physics.
In 1964, he was elected a foreign member of the Royal Netherlands Academy of Arts and Sciences. He was also a member of the American Academy of Arts and Sciences and the American Philosophical Society.
Family
On 14 March 1940, Bloch married Lore Clara Misch (1911–1996), a fellow physicist working on X-ray crystallography, whom he had met at an American Physical Society meeting. They had four children, twins George Jacob Bloch and Daniel Arthur Bloch (born 15 January 1941), son Frank Samuel Bloch (born 16 January 1945), and daughter Ruth Hedy Bloch (born 15 September 1949).
Bloch died on 10 September 1983 in Zurich at the age of 77. In 2025 Bloch's family donated his Nobel Prize medal to CERN.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1896 2026-03-15 00:05:54
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2459) Edward Mills Purcell
Gist:
Life
Edward Mills Purcell was born in Taylorville, Illinois. His father worked for a telephone company. Purcell studied electrical engineering at Purdue University in Indiana and physics at Harvard University. During World War II Purcell worked on the development of radar at MIT, but he returned afterwards to Harvard, where he did his Nobel Prize-awarded work and continued to work for the rest of his career. Purcell was married and had two sons.
Work
Protons and neutrons in nuclei act like small, rotating magnets. Atoms and molecules therefore align in a magnetic field. Radio waves can disturb their direction of rotation, but only in certain stages, in accordance with quantum mechanics. When the atoms return to their original positions, they emit electromagnetic radio waves with frequencies characteristic of different elements and isotopes. In 1946, Edward Purcell and Felix Bloch developed methods for precise measurement, making it possible to study different materials’ compositions.
Summary
E.M. Purcell (born Aug. 30, 1912, Taylorville, Ill., U.S.—died March 7, 1997, Cambridge, Mass.) was an American physicist who shared, with Felix Bloch of the United States, the Nobel Prize for Physics in 1952 for his independent discovery (1946) of nuclear magnetic resonance in liquids and in solids. Nuclear magnetic resonance (NMR) has become widely used to study the molecular structure of pure materials and the composition of mixtures.
During World War II Purcell headed a group studying radar problems at the Radiation Laboratory of the Massachusetts Institute of Technology, Cambridge. In 1946 he developed his NMR detection method, which was extremely accurate and a major improvement over the atomic-beam method devised by the American physicist Isidor I. Rabi.
Purcell became professor of physics at Harvard University in 1949 and in 1952 detected the 21-centimetre-wavelength radiation emitted by neutral atomic hydrogen in interstellar space. Such radio waves had been predicted by the Dutch astronomer H.C. van de Hulst in 1944, and their study enabled astronomers to determine the distribution and location of hydrogen clouds in galaxies and to measure the rotation of the Milky Way. In 1960 Purcell became Gerhard Gade professor at Harvard, and in 1979 he received the National Medal of Science. In 1980 he became professor emeritus.
Details
Edward Mills Purcell (August 30, 1912 – March 7, 1997) was an American physicist who shared the 1952 Nobel Prize for Physics for his independent discovery (published 1946) of nuclear magnetic resonance in liquids and in solids. Nuclear magnetic resonance (NMR) has become widely used to study the molecular structure of pure materials and the composition of mixtures. Friends and colleagues knew him as Ed Purcell.
Biography
Born and raised in Taylorville, Illinois, Purcell received his BSEE in electrical engineering from Purdue University, followed by his M.A. and Ph.D. in physics from Harvard University. He was a member of the Alpha Xi chapter of the Phi Kappa Sigma fraternity while at Purdue. After spending the years of World War II working at the MIT Radiation Laboratory on the development of microwave radar, Purcell returned to Harvard to do research. In December 1945, he discovered nuclear magnetic resonance (NMR) with his colleagues Robert Pound and Henry Torrey. NMR provides scientists with an elegant and precise way of determining chemical structure and properties of materials, and is widely used in physics and chemistry. It also is the basis of magnetic resonance imaging (MRI), one of the most important medical advances of the 20th century. For his discovery of NMR, Purcell shared the 1952 Nobel Prize in physics with Felix Bloch of Stanford University.
Purcell also made contributions to astronomy as the first to detect radio emissions from neutral galactic hydrogen (the famous 21 cm line due to hyperfine splitting), affording the first views of the spiral arms of the Milky Way. This observation helped launch the field of radio astronomy, and measurements of the 21 cm line are still an important technique in modern astronomy. He has also made seminal contributions to solid state physics, with studies of spin-echo relaxation, nuclear magnetic relaxation, and negative spin temperature (important in the development of the laser). With Norman F. Ramsey, he was the first to question the CP symmetry of particle physics.
Purcell was the recipient of many awards for his scientific, educational, and civic work. He served as science advisor to Presidents Dwight D. Eisenhower, John F. Kennedy, and Lyndon B. Johnson. He was president of the American Physical Society, and a member of the American Philosophical Society, the National Academy of Sciences, and the American Academy of Arts and Sciences. He was awarded the National Medal of Science in 1979, and the Jansky Lectureship before the National Radio Astronomy Observatory. Purcell was also inducted into his Fraternity's (Phi Kappa Sigma) Hall of Fame as the first Phi Kap ever to receive a Nobel Prize.
Purcell was the author of the innovative introductory text Electricity and Magnetism. The book, a Sputnik-era project funded by an NSF grant, was influential for its use of relativity in the presentation of the subject at this level. The 1965 edition, now freely available due to a condition of the federal grant, was originally published as a volume of the Berkeley Physics Course. The book is also in print as a commercial third edition, as Purcell and Morin. Purcell is also remembered by biologists for his famous lecture "Life at Low Reynolds Number", in which he explained forces and effects dominating in limiting flow regimes (often at the micro scale). He also emphasized the time-reversibility of low Reynolds number flows with a principle referred to as the Scallop theorem.
Purcell died on March 7, 1997, in Cambridge, Massachusetts, aged 84.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1897 2026-03-16 00:03:34
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2460) Archer Martin
Gist:
Work
When a drop of a liquid containing a mixture of various substances is placed on paper, the liquid begins to spread out on the paper. The various substances in the mixture spread at different speeds, however, which gives rise to marks on the paper with different colors. In the 1940s Archer Martin and Richard Synge used this and similar phenomena in gas mixtures, for example, to develop different types of chromatography—methods for separating substances in mixtures and for determining the composition of mixtures.
Summary
A.J.P. Martin (born March 1, 1910, London, England —died July 28, 2002, Llangarron, Herefordshire) was a British biochemist who was awarded (with R.L.M. Synge) the Nobel Prize for Chemistry in 1952 for development of paper partition chromatography, a quick and economical analytical technique permitting extensive advances in chemical, medical, and biological research.
Martin obtained a Ph.D. from the University of Cambridge in 1936 and worked as a research chemist for the Wool Industries Research Association in Leeds from 1938 to 1946. He then became head of biochemical research at the Boots Pure Drug Company, Nottingham, and held the post until 1948, when he was appointed to the staff of the British Medical Research Council. From 1959 to 1970 he was director of Abbotsbury Laboratories, Ltd. Martin also taught at the University of Houston in Texas (1974–79).
Martin and Synge invented paper partition chromatography in 1944. Partition chromatography depends on the partition, or distribution, of each component of a mixture between two immiscible liquids. One of the liquids is held stationary by strong adsorption on the surface of a finely divided solid while the other flows through the interstices of the solid particles. Any substance that preferentially dissolves in the mobile liquid is more rapidly transported in the direction of flow than is a substance that has greater affinity for the stationary liquid. In 1953 Martin and A.T. James helped perfect gas chromatography, the separation of chemical vapours by differential absorption on a porous solid.
Details
Archer John Porter Martin (1 March 1910 – 28 July 2002) was a British chemist who shared the 1952 Nobel Prize in Chemistry for the invention of partition chromatography with Richard Synge.
Early life
Martin's father was a GP. Martin was educated at Bedford School, and Peterhouse, Cambridge.
Career
Working first in the Physical Chemistry Laboratory, he moved to the Dunn Nutritional Laboratory, and in 1938 moved to Wool Industries Research Institution in Leeds. He was head of the biochemistry division of Boots Pure Drug Company from 1946 to 1948, when he joined the Medical Research Council. There, he was appointed head of the physical chemistry division of the National Institute for Medical Research in 1952, and was chemical consultant from 1956 to 1959.
He specialised in biochemistry, in some aspects of vitamins E and B2, and in techniques that laid the foundation for several new types of chromatography. He developed partition chromatography whilst working on the separation of amino acids, and later developed gas-liquid chromatography with Anthony T. James. Amongst many honours, he received his Nobel Prize in 1952.
After his retirement from the University of Sussex, he was visiting professor at both the University of Houston in Texas and the EPFL (École Polytechnique Fédérale de Lausanne) in Switzerland.
He published far fewer papers than the typical Nobel winners—only 70 in all—but his ninth paper contained the work that would eventually win him the Nobel Prize. The University of Houston dropped him from its chemistry faculty in 1979 (when he was 69 years old) because he was not publishing enough.
Awards
Archer Martin shared the 1952 Nobel Prize in Chemistry for the invention of partition chromatography with Richard Synge.
Archer Martin's 1954 paper with Anthony T. James, "Gas-Liquid Chromatography: A Technique for the Analysis and Identification of Volatile Materials" reported the discovery of gas-liquid chromatography. This was performed at the National Institute for Medical Research in Mill Hill, London, which became the Francis Crick Institute in 2016. It was honoured by a Citation for Chemical Breakthrough Award from the Division of History of Chemistry of the American Chemical Society presented in 2016 to the Francis Crick Institute.
Martin was elected a Fellow of the Royal Society in 1950, and made a CBE in 1960.
Personal life
In 1943 he married Judith Bagenal (1918–2006), and together they had two sons and three daughters. In the last years of his life he suffered from Alzheimer's disease.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1898 2026-03-17 00:03:41
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2461) Richard Laurence Millington Synge
Gist:
Work
When a drop of a liquid containing a mixture of various substances is placed on paper, the liquid begins to spread out on the paper. The various substances in the mixture spread at different speeds, however, which gives rise to marks on the paper with different colors. In the 1940s Richard Synge and Archer Martin used this and similar phenomena in gas mixtures, for example, to develop different types of chromatography—methods for separating substances in mixtures and for determining the composition of mixtures.
Summary
R.L.M. Synge (born Oct. 28, 1914, Liverpool, Eng.—died Aug. 18, 1994, Norwich, Norfolk) was a British biochemist who in 1952 shared the Nobel Prize for Chemistry with A.J.P. Martin for their development of partition chromatography, notably paper chromatography.
Synge studied at Winchester College, Cambridge, and received his Ph.D. at Trinity College there in 1941. He spent his entire professional career conducting research, initially with Martin under the auspices of the Wool Industries Research Association, Leeds (1941–43). The two men developed partition chromatography, a technique that is used to separate mixtures of closely related chemicals such as amino acids for identification and further study. Synge used paper chromatography to work out the exact structure of the simple protein molecule gramicidin S, which helped to pave the way for the English biochemist Frederick Sanger’s elucidation of the structure of the insulin molecule.
Synge did research at the Lister Institute of Preventive Medicine, London (1943–48), and at the Rowett Research Institute, near Aberdeen, Scot. (1948–67). He became a biochemist at the Food Research Institute, Norwich (1967–76), and was also an honorary professor of biological sciences at the University of East Anglia (1968–84).
Details
Richard Laurence Millington Synge (28 October 1914 – 18 August 1994) was a British biochemist, and shared the 1952 Nobel Prize in Chemistry for the invention of partition chromatography with Archer Martin.
Life
Richard Laurence Millington Synge was born in West Kirby on 28 October 1914, the son of Lawrence Millington Synge, a Liverpool stock-broker, and his wife, Katherine C. Swan.
Synge was educated at the Old Hall in Wellington, Shropshire and at Winchester College. He then studied Chemistry at Trinity College, Cambridge.
He spent his entire career in research, at the Wool Industries Research Association, Leeds (1941–1943), Lister Institute for Preventive Medicine, London (1943–1948), Rowett Research Institute, Aberdeen (1948–1967), and Food Research Institute, Norwich (1967–1976).
It was during his time in Leeds that he worked with Archer Martin, developing partition chromatography, a technique used in the separation mixtures of similar chemicals, that revolutionised analytical chemistry. Between 1942 and 1948 he studied peptides of the protein group gramicidin, work later used by Frederick Sanger in determining the structure of insulin. In March 1950 he was elected a Fellow of the Royal Society for which his candidature citation read:
Distinguished as a biochemist. Was the first to show the possibility of using counter-current liquid-liquid extraction in the separation of N-acetylamino acids. In collaboration with A.J.P. Martin this led to the development of partition chromatography, which they have applied with conspicuous success in problems related to the composition and structure of proteins, particularly wool keratin. Synge's recent work on the composition and structure of gramicidins is outstanding and illustrates vividly the great advances in technique for which he and Martin are responsible.
— "Library and Archive catalogue". Royal Society. Archived from the original on 27 July 2011. Retrieved 24 October 2010.
In 1963 he was elected a Fellow of the Royal Society of Edinburgh. His proposers were Magnus Pyke, Andrew Phillipson, Sir David Cuthbertson and John Andrew Crichton.
He was for several years the treasurer of the Chemical Information Group of the Royal Society of Chemistry, and was an honorary Professor in Biological Sciences at the University of East Anglia from 1968 to 1984. He was awarded an honorary Doctor of Science (ScD) from the University of East Anglia in 1977, and an honorary doctorate from the Faculty of Mathematics and Science at Uppsala University, Sweden in 1980.
Personal life
In 1943 Synge married Ann Davies Stephen (1916–1997). Ann Stephen was the daughter of psychologist Karin Stephen and psychoanalyst Adrian Stephen. Ann's sister Judith (1918–1972) was married to documentary artist and photographer Nigel Henderson.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1899 2026-03-18 00:14:54
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2462) Selman Waksman
Gist:
Work
After Robert Koch discovered that tuberculosis is caused by a bacterium, the hunt for a cure began. In 1939 Selman Waksman and colleagues began systematic studies of how microorganisms in soil affect tubercle bacteria. They found that their growth was impeded by another bacterium, Streptomyces grisues. In 1943 Waksman's colleague, Albert Schatz, isolated streptomycin from this bacterium, which proved an effective medicine against tuberculosis.
Summary
Selman Abraham Waksman (born July 22, 1888, Priluka, Ukraine, Russian Empire [now Pryluky, Ukraine]—died August 16, 1973, Hyannis, Massachusetts, U.S.) was a Ukrainian-born American biochemist who was one of the world’s foremost authorities on soil microbiology. After the discovery of penicillin, he played a major role in initiating a calculated, systematic search for antibiotics among microbes. His screening methods and consequent co-discovery of the antibiotic streptomycin, the first specific agent effective in the treatment of tuberculosis, brought him the 1952 Nobel Prize for Physiology or Medicine.
A naturalized U.S. citizen (1916), Waksman spent most of his career at Rutgers University, New Brunswick, New Jersey, where he served as professor of soil microbiology (1930–40), professor of microbiology and chairman of the department (1940–58), and director of the Rutgers Institute of Microbiology (1949–58). During his extensive study of the actinomycetes (filamentous, bacteria-like microorganisms found in the soil), he extracted from them antibiotics (a term he coined in 1941) valuable for their killing effect not only on gram-positive bacteria, such as the tubercle bacillus (Mycobacterium tuberculosis, which unlike other gram-positive microbes is insensitive to penicillin), but also on gram-negative bacteria, such as the organisms that cause cholera (Vibrio cholerae) and typhoid fever (Salmonella typhi).
In 1940 Waksman, along with his graduate student H. Boyd Woodruff, isolated actinomycin from soil bacteria. Although the substance was effective against strains of gram-negative and gram-positive bacteria, including M. tuberculosis, it was extremely toxic when given to test animals. Four years later Waksman and graduate students Albert Schatz and Elizabeth Bugie published a paper describing their discovery of the relatively nontoxic streptomycin, which they extracted from the actinomycete Streptomyces griseus. They found that the antibiotic exercised repressive influence on tuberculosis. In combination with other chemotherapeutic agents, streptomycin has become a major factor in controlling the disease. Waksman also isolated and developed several other antibiotics, including neomycin, that have been used in treating many infectious diseases of humans, domestic animals, and plants.
Among Waksman’s books are Principles of Soil Microbiology (1927), regarded as one of the most exhaustive works on the subject, and My Life with the Microbes (1954), an autobiography.
Details
Selman Abraham Waksman (July 22, 1888 – August 16, 1973) was a Russian-born American inventor, biochemist and microbiologist, whose research into the decomposition of organisms that live in soil enabled the discovery of streptomycin and several other antibiotics. For his work he won the 1952 Nobel Prize in Physiology or Medicine.
Waksman emigrated to the United States in 1910 and became a naturalized U.S. citizen in 1916. A professor of biochemistry and microbiology at Rutgers University for four decades, he discovered several antibiotics (and introduced the modern sense of that word to name them), and he introduced procedures that have led to the development of many others. The proceeds earned from the licensing of his patents funded a foundation for microbiological research, which established the Waksman Institute of Microbiology located at the Rutgers University Busch Campus in Piscataway, New Jersey (USA). After receiving the Nobel Prize, Waksman and his foundation later were sued by Albert Schatz, one of his Ph.D. students and the discoverer of streptomycin, for minimizing Schatz's role in the discovery.
In 2005, Waksman was granted an ACS National Historic Chemical Landmark in recognition of the significant work of his lab in isolating more than 15 antibiotics, including streptomycin, which was the first effective treatment for tuberculosis.
Early life and education
Selman Waksman was born on July 22 [O.S. July 8] 1888, to Jewish parents, in Nova Pryluka, Kiev Governorate, Russian Empire, now Vinnytsia Oblast, Ukraine. He was the son of Fradia (London) and Jacob Waksman. In 1910, shortly after receiving his diploma from the Fifth Gymnasium in Odessa, he immigrated to the United States and became a naturalized American citizen in 1916.
Waksman attended Rutgers College (now Rutgers University), where he graduated in 1915 with a Bachelor of Science in agriculture. He continued his studies at Rutgers, receiving a Master of Science the following year, in 1916. During his graduate study, he worked under J. G. Lipman at the New Jersey Agricultural Experiment Station at Rutgers performing research in soil bacteriology. Waksman spent some months in 1915–1916 at the United States Department of Agriculture in Washington, DC under Charles Thom, studying soil fungi. He was appointed as a research fellow at the University of California, Berkeley, and in 1918 he was awarded his doctor of philosophy in biochemistry.
Career
He joined the faculty at Rutgers University in the Department of Biochemistry and Microbiology.
At Rutgers, Waksman's team discovered several antibiotics, including actinomycin, clavacin, streptothricin, streptomycin, grisein, neomycin, fradicin, candicidin, candidin. Waksman co-discovered streptomycin with Albert Schatz. Streptomycin was the first effective drug against gram-negative bacteria and the first antibiotic used to cure tuberculosis. Waksman is credited with coining the term antibiotics to describe antibacterials derived from other living organisms, for example penicillin, though the term was used by the French dermatologist François Henri Hallopeau, in 1871 to describe a substance opposed to the development of life.
In 1931, Waksman organized the division of Marine Bacteriology at the Woods Hole Oceanographic Institution (WHOI) in addition to his task at Rutgers. He was appointed a marine bacteriologist there and served until 1942. He was elected a trustee at WHOI and finally a Life Trustee.
In 1951, using half of his patent royalties, Waksman created the Waksman Foundation for Microbiology. At a meeting of the board of trustees of the foundation, held in July 1951, he urged the building of a facility for work in microbiology, named the Waksman Institute of Microbiology, which is located on the Busch Campus of Rutgers University in Piscataway, New Jersey. The foundation's first president, Waksman, was succeeded in this position by his son, Byron H. Waksman, from 1970 to 2000.
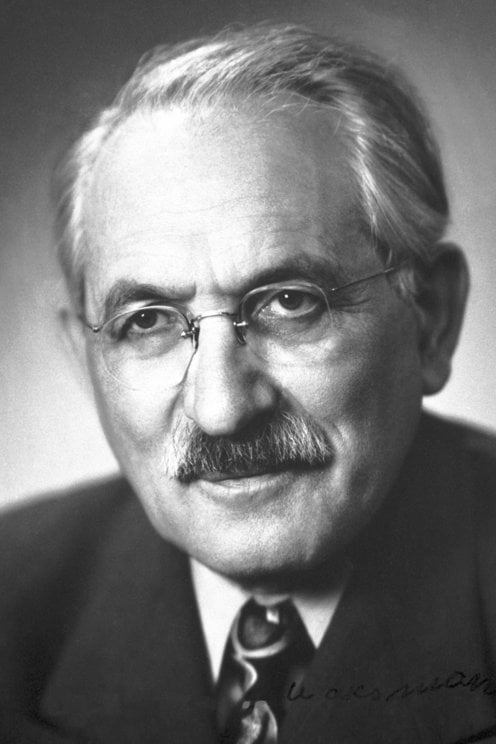
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#1900 2026-03-19 00:26:41
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Re: crème de la crème
2463) Ernest Walton
Gist:
Life
Ernest Walton was born into a clergyman’s family in southern Ireland. After studying at the international school in Belfast and Trinity College in Dublin, in 1927 he received a scholarship to work at the Cavendish Laboratory in Cambridge. There, Walton conducted his Nobel Prize-winning work at the beginning of the 1930s. In 1934 Walton returned to Trinity College in Dublin, where he remained for the rest of his active life. Walton married the same year and had two sons and two daughters.
Work
Ernest Rutherford used alpha particles from radioactive elements to study nuclear reactions and used his findings to convert nitrogen into oxygen. However, only a very few nuclear reactions could be achieved using alpha particles. Ernest Walton and John developed a device, an accelerator, to generate more penetrating radiation. Using a strong electric field, protons were accelerated to high velocities. In 1932, they bombarded lithium with protons, causing their nuclei to split and producing two alpha particles.
Summary
Ernest Thomas Sinton Walton (born Oct. 6, 1903, Dungarvan, County Waterford, Ire.—died June 25, 1995, Belfast, N.Ire.) was an Irish physicist, corecipient, with Sir John Douglas of England, of the 1951 Nobel Prize for Physics for the development of the first nuclear particle accelerator, known as the math-Walton generator.
After studying at the Methodist College, Belfast, and graduating in mathematics and experimental science from Trinity College, Dublin (1926), Walton went in 1927 to Trinity College, Cambridge, where he was to work with John in the Cavendish Laboratory under Lord Rutherford until 1934. In 1928 he attempted two methods of high-energy particle acceleration. Both failed, mainly because the available power sources could not generate the necessary energies, but his methods were later developed and used in the betatron and the linear accelerator. Then in 1929 John and Walton devised an accelerator that generated large numbers of particles at lower energies. With this device in 1932 they disintegrated lithium nuclei with protons, the first artificial nuclear reaction not utilizing radioactive substances.
After gaining his Ph.D. at Cambridge, Walton returned to Trinity College, Dublin, in 1934, where he remained as a fellow for the next 40 years and a fellow emeritus thereafter. He was Erasmus Smith professor of natural and experimental philosophy from 1946 to 1974 and chairman of the School of Cosmic Physics at the Dublin Institute for Advanced Studies after 1952.
Details
Ernest Thomas Sinton Walton (6 October 1903 – 25 June 1995), commonly abbreviated as E. T. S Walton, was an Irish experimental physicist and academic. He shared the 1951 Nobel Prize in Physics with John "for their pioneer work on the transmutation of atomic nuclei by artificially accelerated atomic particles." According to their Nobel Prize speech: "Thus, for the first time, a nuclear transmutation was produced by means entirely under human control."
Walton was a key member of the nuclear physics faculty at the University of Cambridge, where he worked with math and Ernest Rutherford. He then spent the majority of his career in Ireland, after returning from England in 1934. He remained active as a member of the teaching faculty at Trinity College Dublin, where he served as the Erasmus Smith's Professor of Natural and Experimental Philosophy from 1946 until his retirement in 1974, after which he continued to be associated with the physics department at the college. Along with William Rowan Hamilton, Walton is regarded as one of the most influential Irish physicists.
Early life and education
Ernest Thomas Sinton Walton was born on 6 October 1903 in Dungarvan, Ireland, the son of John Walton (1874–1936), a Methodist minister from Cloughjordan, and Anna Sinton (1874–1906) from Richhill. In those days, a general clergyman's family moved once every three years, and this practice carried Ernest and his family, while he was a small child, to Rathkeale, County Limerick (where his mother died), and to County Monaghan.
Walton attended day schools in counties Down and Tyrone, and at Wesley College Dublin before becoming a boarder at Methodist College Belfast in 1915, where he excelled in science and mathematics.
In 1922, Walton won scholarships to Trinity College Dublin for the study of mathematics and science, and would go on to be elected a Foundation Scholar in 1924. He was awarded bachelor's and master's degrees from Trinity in 1926 and 1927, respectively. During these years at college, he received numerous prizes for excellence in physics and mathematics (seven prizes in all), including the Foundation Scholarship in 1924. After graduating in 1927, he was awarded an 1851 Research Fellowship from the Royal Commission for the Exhibition of 1851 and was accepted as a research student at Trinity College, Cambridge, under the supervision of Ernest Rutherford, Director of the Cavendish Laboratory. At the time there were four Nobel Prize laureates on the staff in the Cavendish Laboratory and a further five were to emerge, including Walton and John Walton received his Ph.D. in 1931, and remained at Cambridge as a researcher until 1934.
Research
During the early 1930s, Walton and John collaborated to build an apparatus that split the nuclei of lithium atoms by bombarding them with a stream of protons accelerated inside a high-voltage tube (700 kilovolts). The splitting of the lithium nuclei produced helium nuclei. They went on to use boron and carbon as targets for their 'disintegration' experiments, and to report induced radioactivity. These experiments provided verification of theories about atomic structure that had been proposed earlier by Rutherford, George Gamow, and others. The successful apparatus—a type of particle accelerator, now called the math–Walton generator—helped to usher in an era of particle accelerator-based experimental nuclear physics. It was this research at Cambridge in the early 1930s that won Walton and John the Nobel Prize in Physics in 1951.
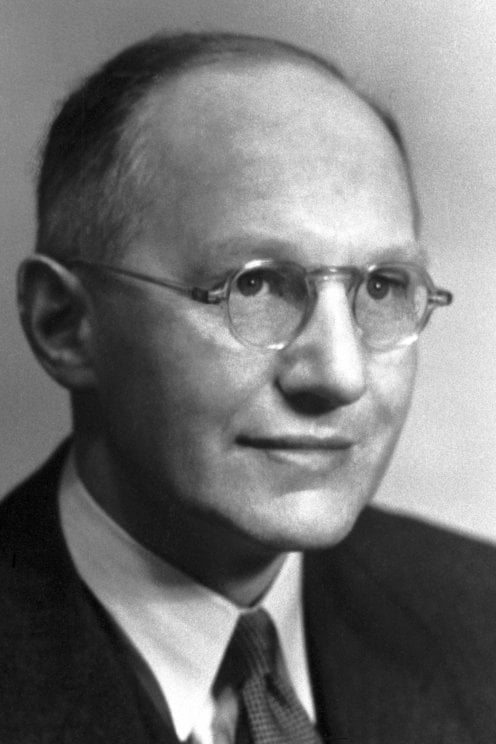
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline