Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
#51 Jokes » Miscellaneous Food Jokes - IV » 2026-03-08 00:06:46
- Jai Ganesh
- Replies: 0
Q: What kind of witch crafts food?
A: A sand-witch.
* * *
Q: How does butter play baseball?
A: It butters up.
* * *
Q: Why do they call it Fast Food?
A: Because if you don't eat it really fast, you might actually taste it.
* * *
Q: What happens when you use pickles for a ping pong game?
A: You get a volley of the Dills.
* * *
#52 Dark Discussions at Cafe Infinity » Come and Go Quotes » 2026-03-08 00:06:19
- Jai Ganesh
- Replies: 0
Come and Go Quotes
1. Yes, you can lose somebody overnight, yes, your whole life can be turned upside down. Life is short. It can come and go like a feather in the wind. - Shania Twain
2. I don't give up. I'm a plodder. People come and go, but I stay the course. - Kevin Costner
3. I like having the dough to come and go as I please. - Bruce Willis
4. People say that artists come and go but let me tell you I am not an artist. I am first a human being and if you are a good human being, you have achieved everything no matter how big actor you are. - Dharmendra
5. People come and go in your life. It is up to you choose how you want to associate with that person. It is up to you to learn and imbibe things from such a person. - Rekha
6. The role has to excite me as an actor. It is like a train journey: you know where you have to reach, and all things that happen in between are like stations that come and go. Of course, there are films like 'Welcome,' which I did only for money. - Nana Patekar
7. Money and other tangibles will come and go; they shouldn't be the foundation of your marriage. - Hema Malini
8. I see people every day who think they're the be all and end all of the industry. I've seen so many people come and go, but the industry doesn't revolve around one person. - Farah Khan.
#53 This is Cool » Detergent » 2026-03-07 17:57:05
- Jai Ganesh
- Replies: 0
Detergent
Gist
A detergent is a cleaning agent, often called a "surfactant," that uses molecules with both water-loving (polar) heads and oil-loving (non-polar) tails to lift dirt, grease, and oil from surfaces, suspending them in water so they can be rinsed away. Unlike traditional soaps, detergents are synthetic, work effectively in hard water, and are formulated into various products like laundry liquids, dishwasher tabs, and shampoos, containing additional ingredients for foaming, softening, or scent
Detergent products on the market can be categorized into powder detergents, liquid detergents, detergent bars, detergent pods, etc. The primary purpose is to remove dirt, stains, sweat, oils, pathogens, and other contaminants from fabrics, leaving them clean, fresh, and often pleasantly scented.
Summary
A detergent is a product for cleaning that contains surfactants plus other components. Detergents comprise surfactants as main functional components to remove hydrophobic grease or dirt by dispersing them in water. They often further comprise water (to facilitate application), builders (to soften water), enzymes (for breaking down proteins, fats, or starches), and dyes or fragrances (to improve the user's sensory experience).
Common surfactants used in detergents are alkylbenzene sulfonates, which are soap-like compounds that are more soluble than soap in hard water, because the polar sulfonate is less likely than the polar carboxylate of soap to bind to calcium and other ions found in hard water.
Definitions
The word detergent is derived from the Latin adjective detergens, from the verb detergere, meaning to wipe or polish off. Detergent can be defined as a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. However, conventionally, detergent is used to mean synthetic cleaning compounds as opposed to soap (a salt of the natural fatty acid), even though soap is also a detergent in the true sense. In domestic contexts, the term detergent refers to household cleaning products such as laundry detergent or dish detergent, which are in fact complex mixtures of different compounds, not all of which are by themselves detergents. Detergency is the ability to remove unwanted substances termed 'soils' from a substrate (e.g., clothing).
Details
Key Takeaways:
* Laundry detergents clean fabrics. Surfactants are the key ingredient that allows detergents to remove dirt and stains by interacting with both oil and water.
* Modern detergents contain a mixture of surfactants, enzymes and other ingredients to tackle various stains and fabric care needs.
* The choice between powder and liquid detergents often comes down to personal preference, although environmental considerations and packaging waste are increasingly important factors for consumers.
Nothing beats the feeling of putting on a clean T-shirt, especially after a shower. It leaves you feeling fresh and ready to take on the day. And where do these clean T-shirts come from? Ah, yes -- the laundry, that household chore that never seems to go off of your to-do list. Even the numbers agree: The average American family does about 300 loads of laundry per year [source: Wall Street Journal].
You may find yourself constantly putting clothes and sheets in the wash because, put simply, people are dirty. We sweat, shed skin cells and come into contact with food, dirt and many more particles every day. Consequently, we need a way to effectively get clothes and fabrics clean to maintain personal hygiene and keep up the appearance of garments. But what exactly is going on in that washing machine to get our clothes and fabrics clean? The secret is laundry detergent.
Prior to the invention of laundry detergents, Americans used soap flakes to wash clothes. However, in the 1930s, the first laundry detergent, Dreft by Procter & Gamble, hit the market. Later, in 1943, Procter & Gamble produced Tide, which could get out tougher stains due to the use of some very unique chemical ingredients. Since the 1930s and 1940s, laundry detergent has become a household necessity that is used around the world to clean fabrics. So how exactly do laundry detergents get your clothes clean? In this article we are going to get to the bottom of what happens when detergents enter your washing machine.
Surfactants: Laundry Detergent's Cleaning Power
All laundry detergent ingredients have a job to do, but the one group that's really crucial to getting your clothes clean are surfactants. The word surfactant stems from the combination of words "surface-active agents." Surface-active agents get their name from their unique chemical structure, which allows them to interact with two different types of surfaces, such as oil and water. The tail of a surfactant molecule is hydrophobic, or not attracted to water. What the hydrophobic end is attracted to is grease and dirt. The head of the surfactant molecule, on the other hand, is hydrophilic -- it's attracted to water [source: Silberberg].
So when a greasy piece of clothing is immersed in water with detergents containing surfactant, the tail of the surfactant molecules attach to the grease, and the head end of the molecule is attracted to the water. When the washing machine agitates the clothes, the molecules form tiny spheres, which stay suspended in the water and are rinsed away when the water is drained. Therefore, the prime benefit of surfactants is their ability to draw grime out of clothing while making sure it doesn't return to the fabrics.
Essentially, there are four main types of surfactants, with the first three used the most in laundry detergents, and their actions depend on their interactions with ions. Ions are charged particles due to the gain or loss of electrons. Ions can be positive such as calcium, Ca2+, or negative such as chloride, Cl-.
* Anionic surfactants are negatively charged in solution. However, they do not work as well by themselves in hard water. This is because hard water has many positively charged ions presents such as calcium (Ca2+) and magnesium (Mg2+). Since anionic surfactants are negative they are attracted to the positive ions and bind, making them unable to bind to other molecules in solution.
* Nonionic surfactants have no charge. Therefore, they are not as easily impaired under hard water conditions, since they are not attracted to the positive ions.
* Cationic surfactants are positively charged in solution. They help the anionic surfactant molecules pack in at the water/dirt interface thereby allowing the anionic surfactants to pull more dirt away.
* Amphoteric or zwitterionic surfactants are both positively and negatively charged. These surfactants are very mild and are often found in gentler cleansers such as hand soaps, shampoos and cosmetics. [source: Silberberg].
Additional Components of Laundry Detergent
Although surfactants are at the heart of laundry detergent's ability to clean fabrics, other ingredients can help detergents clean better, brighten clothes or smell better. As described previously, some types of surfactants typically do not work well in hard water due to the excess positive ions present. Additives called builders can help detergents to work better under hard water conditions. Builders accomplish this feat by removing calcium (Ca2+) and magnesium (Mg2+) ions in hard water by binding to them. This allows the surfactants, especially anionic surfactants, to bind to more grime, rather than the positively charged ions in the wash water. Builders also are bases, so they work to neutralize acid and can help disrupt chemical bonds. Another benefit of adding builders to laundry detergents is that manufacturers can use less surfactant, since the builders make the surfactant more efficient. Some examples of builders include sodium tripolyphosphate (STTP) and zeolites [source: EPA].
Detergents can also include components that make clothes whiter or brighter. The most common whitening agents are bleaches. Bleaches contain peroxides, which can oxidize fabrics [source: EPA]. Fluorescent whiteners and brighteners are also added to some laundry detergents because they minimize the yellowing of fabrics. These additives work by absorbing ultraviolet light and emitting back visible blue light, which can mask the yellow that may make colors appear faded and whites appear dingy.
Enzymes are naturally occurring biologic agents present in many detergents in varying concentrations. These enzymes are typically classified into the following categories and are similar to the enzymes used by your body to digest food:
Proteases: help break down proteins
Lipases: help break down fat
Amylases: help break down starches [source: Basketter]
These enzymes help break down food particles that are present on clothing by catalyzing, or speeding up, the decomposition process. A point to consider is that enzymes are biological products that can break down over time. Therefore, detergents can also contain enzyme stabilizers, which protect the enzymes and help them function.
Some other components include fragrance and coloring, which give laundry detergents their distinctive scents and appearance. Detergents sometimes contain trace amounts of dye, which is not enough to dye your actual clothing. However, on top of making your laundry detergent more visually appealing, dyes can show you when there is still detergent left on your clothes after the wash cycle.
Lastly, fillers help dilute and distribute the active ingredients to their proper dosages. Powder and liquid detergents use different fillers. The major filler in powder detergents is sodium sulphate, which provides the granular powdery texture. The primary filler in liquid detergents is water.
Powder vs. Liquid Detergents
Laundry detergent manufacturers have come a long way since the first box of Tide was produced more than 60 years ago. Currently, the two main types of laundry detergent are powders and liquids. For the most part, powder and liquid detergents share the same active ingredients except for the filler used. Additionally, powder and liquid detergents both have pros and cons, and since they have similar cleaning power, people usually choose which type to use based on personal preference.
Here are some of the advantages and disadvantages of using powdered detergents:
Pro: They're generally cheaper.
Pro: The cardboard packaging is more eco-friendly.
Con: Some people think they don't dissolve as well in water. This may have been a problem with some of the first powdered detergents, but these days, most powders are designed to readily dissolve in water.
Con: Sodium sulphate can wreak havoc on septic systems.
Con: Powders contain more chemicals compared with liquids, due to the filler.
People may or may not use liquid detergents for an entirely different set of reasons:
Pro: The detergent is already pre-dissolved.
Pro: You can pre-treat stains by pouring it directly onto clothes.
Con: They're usually more expensive than powdered detergent.
Con: They have plastic packaging, which is less eco-friendly.
Environmental Considerations with Laundry Detergent
Even though detergents do a tremendous job of getting rid of the dirt and grime in our fabrics, at what cost does this come? Considering the toxicities of their chemical ingredients and carbon cost of production, it's not surprising that some people have concerns about the impacts of laundry detergents on the environment.
Their carbon footprint alone is significant by many people's standards. Carbon footprints are an indicator of the amount of carbon dioxide (CO2) produced while making, shipping and using a product. According to the Wall Street Journal, the carbon footprint of using UK detergent brand Tesco, varies from 1.3 pounds (0.6 kilograms) to 1.9 pounds (0.9 kilograms) per load, depending on the form of the detergent that's used. To put this in perspective, it is estimated that for every mile an average car travels, 1 pound (0.5 kilograms) of CO2 is emitted. Recall that American families on average do 300 loads of laundry per year. This means that the carbon footprint of laundry detergents for one year of laundry is approximately 480 pounds (218 kilograms) per year, or about 10 pounds (4.5 kilograms) per week. So, while this may not seem like a lot, especially if your car produces about 5 tons of CO2 per year, this number only reflects the laundry detergent. It does not factor in the extra energy requirements of running the washer and dryer [source: Wall Street Journal].
Now, add to that the toxic effects of the chemical components in detergents. According to the EPA, some of the major concerns about the chemical ingredients used in laundry detergents include the following:
* Toxicity to aquatic organisms and algae
* Persistence in the environment
* Eutrophication of fresh water, particularly by phosphate-based detergents (now, phosphates have been replaced by zeolites which may be alleviating this problem)
* Health problems in people, such as cancer [source: EPA]
Another concern relating to laundry detergent is that it can make the wash water acidic, and depending on where that water runs to, it could further impact the environment, having effects similar to acid rain.
Green Laundry Detergent Options
Given some of these environmental considerations about laundry detergents, there are some greener options available to today's consumer. Most detergents marketed as environmentally friendly don't include perfumes or dyes, and they're typically phosphate free, biodegradable, and they haven't been tested on animals.
One eco-conscious option is detergent designed to work well in cold water. On average, 80 to 85 percent of the total energy used washing a load of clothes goes to heating up the water [source: Sabaliunas et al.]. Washing in cold water saves energy, which can translate to savings on your household energy bills, too.
Another environmental approach is to use concentrated formulas, which cuts down on packaging and on the amount of water it takes to make the detergent. According to Proctor & Gamble spokeswoman Carol Berning, concentrated detergents require "less plastic for bottles, less corrugated cardboard for crating, and less gasoline used, because we need less trucks to move the shipments" [source: Consumer Reports]. The cold water and concentrated options that different companies manufacture may be one step to greener washing practices. However, even in these forms, the detergents still contain some potentially environmentally hazardous chemicals.
An additional green choice -- for the benefit of the environment and you wallet -- could be making your own laundry detergents. There are a variety of recipes out there, with the common ingredients of water, bar soap, borax and washing soda. Some environmental benefits of making your own laundry detergent are that they typically use fewer chemicals and additives, and they can save on packaging. However, be aware that clothing washed with homemade detergent may also require bleaching, and it may not get stains out as well as some of the commercially produced detergents.
Clearly, detergents are chemically complex products that are continually being improved upon, whether it is boosting their stain-fighting powers or making them greener.
Additional Information
Soap and detergent are substances that, when dissolved in water, possess the ability to remove dirt from surfaces such as the human skin, textiles, and other solids. When soap and water are not available for hand washing or when repeated hand washing compromises the natural skin barrier (e.g., causing scaling or fissures to develop in the skin), hand sanitizers—coming in foam, gel, or liquid form—have been recommended.
The seemingly simple process of cleaning a soiled surface is, in fact, complex and consists of the following physical-chemical steps:
* Wetting of the surface and, in the case of textiles, penetration of the fibre structure by wash liquor containing the detergent. Detergents (and other surface-active agents) increase the spreading and wetting ability of water by reducing its surface tension—that is, the affinity its molecules have for each other in preference to the molecules of the material to be washed.
* Absorption of a layer of the soap or detergent at the interfaces between the water and the surface to be washed and between the water and the soil. In the case of ionic surface-active agents (explained below), the layer formed is ionic (electrically polar) in nature.
* Dispersion of soil from the fibre or other material into the wash water. This step is facilitated by mechanical agitation and high temperature; in the case of hand soap, soil is dispersed in the foam formed by mechanical action of the hands.
* Preventing the soil from being deposited again onto the surface cleaned. The soap or detergent accomplishes this by suspending the dirt in a protective colloid, sometimes with the aid of special additives. In a great many soiled surfaces the dirt is bound to the surface by a thin film of oil or grease. The cleaning of such surfaces involves the displacement of this film by the detergent solution, which is in turn washed away by rinse waters. The oil film breaks up and separates into individual droplets under the influence of the detergent solution. Proteinic stains, such as egg, milk, and blood, are difficult to remove by detergent action alone. The proteinic stain is nonsoluble in water, adheres strongly to the fibre, and prevents the penetration of the detergent. By using proteolytic enzymes (enzymes able to break down proteins) together with detergents, the proteinic substance can be made water-soluble or at least water-permeable, permitting the detergent to act and the proteinic stain to be dispersed together with the oily dirt. The enzymes may present a toxic hazard to some persons habitually exposed.
If detached oil droplets and dirt particles did not become suspended in the detergent solution in a stable and highly dispersed condition, they would be inclined to flocculate, or coalesce into aggregates large enough to be redeposited on the cleansed surface. In the washing of fabrics and similar materials, small oil droplets or fine, deflocculated dirt particles are more easily carried through interstices in the material than are relatively large ones. The action of the detergent in maintaining the dirt in a highly dispersed condition is therefore important in preventing retention of detached dirt by the fabric.
In order to perform as detergents (surface-active agents), soaps and detergents must have certain chemical structures: their molecules must contain a hydrophobic (water-insoluble) part, such as a fatty acid or a rather long chain carbon group, such as fatty alcohols or alkylbenzene. The molecule must also contain a hydrophilic (water-soluble) group, such as ―COONa, or a sulfo group, such as ―OSO3Na or ―SO3Na (such as in fatty alcohol sulfate or alkylbenzene sulfonate), or a long ethylene oxide chain in nonionic synthetic detergents. This hydrophilic part makes the molecule soluble in water. In general, the hydrophobic part of the molecule attaches itself to the solid or fibre and onto the soil, and the hydrophilic part attaches itself to the water.
Four groups of surface-active agents are distinguished:
* Anionic detergents (including soap and the largest portion of modern synthetic detergents), which produce electrically negative colloidal ions in solution.
* Cationic detergents, which produce electrically positive ions in solution.
* Nonionic detergents, which produce electrically neutral colloidal particles in solution.
* Ampholytic, or amphoteric, detergents, which are capable of acting either as anionic or cationic detergents in solution depending on the pH (acidity or alkalinity) of the solution.
The first detergent (or surface-active agent) was soap. In a strictly chemical sense, any compound formed by the reaction of a water-insoluble fatty acid with an organic base or an alkali metal may be called a soap. Practically, however, the soap industry is concerned mainly with those water-soluble soaps that result from the interaction between fatty acids and alkali metals. In certain cases, however, the salts of fatty acids with ammonia or with triethanolamine are also used, as in shaving preparations.

#54 Science HQ » Ulna (Bone) » 2026-03-07 17:10:00
- Jai Ganesh
- Replies: 0
Ulna (Bone)
Gist
The ulna is the longer, larger medial bone of the forearm, running parallel to the radius from the elbow to the wrist. It acts as a stable hinge for the elbow via the olecranon process and facilitates forearm rotation. It is essential for elbow stability, forearm rotation (pronation/supination), and serves as a major site for muscle attachment.
What is the ulna? The ulna is the longer of the two bones in your forearm. It helps you move your arm, wrist and hand. Your ulna also supports lots of important muscles, tendons, ligaments and blood vessels.
Summary
The ulna or ulnar bone (pl.: ulnae or ulnas) is a long bone in the forearm stretching from the elbow to the wrist. It is on the same side of the forearm as the little finger, running parallel to the radius, the forearm's other long bone. Longer and thinner than the radius, the ulna is considered to be the smaller long bone of the lower arm. The corresponding bone in the lower leg is the fibula.
Structure
The ulna is a long bone found in the forearm that stretches from the elbow to the wrist, and when in standard anatomical position, is found on the medial side of the forearm. It is broader close to the elbow, and narrows as it approaches the wrist.
Close to the elbow, the ulna has a bony process, the olecranon process, a hook-like structure that fits into the olecranon fossa of the humerus. This prevents hyperextension and forms a hinge joint with the trochlea of the humerus. There is also a radial notch for the head of the radius, and the ulnar tuberosity to which muscles attach.
Close to the wrist, the ulna has a styloid process.
Details:
Description
The ulna is one of two bones that make up the forearm, the other being the radius. It forms the elbow joint with the humerus and also articulates with the radius both proximally and distally. It is located in the medial forearm when the arm is in the anatomical position. It is the larger of the two forearm bones. Ulna assists in pronation and supination of the forearm and hand.
Structure
The ulna is a long bone larger proximally than distally.
Proximal ulna
The proximal ulna is hook-like in form which articulates with the trochlea of the humerus to create the hinge joint of the Elbow.
The articulation is formed of the olecranon and the coronoid process.
Olecranon
This is a large, curved bony prominence which is accepted into the olecranon fossa, located on the humerus, during elbow extension.
The olecranon forms the upper part of the semi-lunar notch which is a smooth, large depression and articulates with the humeral trochlea during elbow flexion and extension.
Coronoid process
The coronoid process is a horizontal, bony projection which attaches directly onto the ulnar shaft. It is received into the coronoid fossa of the humerus in elbow flexion. The coronoid process also forms the lower part of the semi-lunar notch.
On the lateral side of the coronoid process is the radial notch where the head of the radius sits.
Head of the ulna
The lateral, distal end of the ulna is the head of the ulna. It articulates with the ulnar notch on the radius and with the triangular articular disc in the Wrist Joint.
Shaft of the Ulna
The shaft of ulna is triangular in shape and it consists of three borders and three surfaces. It's width is decreased as it moves towards distal end.
The three surfaces are:
* Anterior surface
* Posterior Surface
* Medial Surface
The three borders are:
* Interosseous Border
* Anterior Border
* Posterior Border
Distal End
The head of Ulna has convex articular surface on its lateral side in order to articulate with ulnar notch of radius. It forms the distal radio-ulnar joint.
The styloid process has attachment of ulnar collateral ligament.
Articulations:
Elbow
The ulna articulates with the humerus at its most proximal point forming the elbow in a hinge joint. It is the trochlea of the humerus which sits in the semi-lunar notch of the ulna to form this joint.
Radio-ulnar joints
The ulna articulates with the radius proximally and distally to produce pronation (from the proximal joint) and supination (from the distal joint) of the forearm.
The ulna also articulates with the radius in a syndesmosis joint via its interosseous membrane. Which runs the length of the shaft of the ulna from the radial notch proximally to the head of the ulna. This also acts to compartmentalise dorsal and volar sides of the forearm.
Muscle attachments:
Olecranon process
Triceps - inserts onto the posterior of the olecranon process.
Anconeus - inserts onto lateral aspect
Flexor Carpi Ulnaris - origin:posterior, also shares an origin from humeral medial epicondyle
Coronoid process
Brachialis - inserts to anterior, inferior coronoid process
Pronator teres - originates medial surface, also from humeral medial epicondyle
Flexor Digitorum Superficialis - originates medial surface, also from humeral medial epicondyle
Shaft of ulna
All of the following arise from the shaft of the ulna:
Flexor Digitorum Profundus
Pronator quadratus
Extensor carpi ulnaris - also shared with lateral epicondyle of humerus
Abductor pollicis longus - also originates from interosseous membrane
Extensor Pollicis Longus - also originates from interosseous membrane
Extensor indicis - also originates from interosseous membrane
Clinical relevance
Like any other joint in the body the ulna can be affected by osteoarthritis in the elbow joint.
Ulnar fractures
Typically occur as a result of fragility and normally following a fall such as a fall on an outstretched hand (FOOSH)
Monteggia fracture - fracture of the proximal 1/3 of the ulnar shaft accompanied by the dislocation of the radial head.
Hume fracture - fracture of the olecranon accompanied by anterior dislocation of the radial head.
Galezzi's fracture - fracture to the distal radius accompanied by ulnar head dislocation at distal radio-ulna joint.
Additional Information
The ulna is one of the bones in your forearm. It helps you move your arm, wrist and hand. If osteoporosis weakens your bones, you have an increased risk for fractures you might not even know about. Talk to a healthcare provider about a bone density test.
Overview:
The ulna is the longer of the two bones in your forearm.
What is the ulna?
The ulna is the longer of the two bones in your forearm. It helps you move your arm, wrist and hand. Your ulna also supports lots of important muscles, tendons, ligaments and blood vessels.
If you experience a fractured (broken) ulna, you might need surgery to repair your bone and physical therapy to help you regain your strength and ability to move.
Your ulna — like all bones — can be affected by osteoporosis.
Because your ulna is connected to so many muscles and nerves, injuries to one can often affect the others.
Function:
What does the ulna do?
Your ulna has several important jobs, including:
* Helping your forearm and wrist move, flex and rotate.
* Holding more than a dozen muscles in place.
* Stabilizing the rest of your arm, wrist and hand.
* Helping your elbow and wrist move.
Anatomy:
Where is the ulna located?
The ulna is one of two bones in your forearm. The other is your radius. The ulna is on the medial (pinkie) side of your forearm.
What does the ulna look like?
The ulna has a notched end where it meets your humerus (upper arm bone), a long shaft in the middle that’s slightly curved and a narrow end that meets your wrist. It’s slightly longer than the radius.
Even though it’s one long bone, your ulna is made up of several parts. These include:
Ulna proximal aspect
The upper (proximal) end of your ulna connects to your humerus. It forms the bony point of your elbow. The ulna’s proximal end (aspect) contains the:
* Olecranon.
* Trochlear notch.
* Coronoid process.
* Radial notch.
* Ulnar tuberosity.
Ulna shaft
The shaft is the long middle portion of the ulna that supports the weight of your forearm and gives it its shape.
Ulna distal aspect
The lower (distal) end of your ulna forms part of your wrist joint. It ends in a rounded bump called the styloid process — the bump above your wrist on your pinkie side. The distal end is sometimes referred to as the head of your ulna.
Your healthcare provider may use all these parts and labels as they describe where you’re having pain or issues. If you ever break your ulna (an ulnar fracture), your provider might use some of these terms to describe where your bone was damaged.
How big is the ulna?
The ulna is the second longest bone in your arm. It’s one of the longest bones in your body. Most adults’ ulna bones are around 11 inches (in.) long.
Conditions and Disorders:
What are the common conditions and disorders that affect the ulna?
The most common issues that affect the ulna are fractures, osteoporosis and damage to the ulnar nerve. Common conditions include:
* Osteoarthritis.
* Ulnar wrist pain.
* Ulnar nerve entrapment.
Ulna fractures
A bone fracture is the medical term for a broken bone. The most common causes of ulna fractures include:
* Sports injuries.
* Fall.
* Car accidents.
Symptoms of a fracture include:
* Pain.
* Swelling.
* Tenderness.
* Inability to move your arm like you usually can.
* Bruising or discoloration.
* A deformity or bump that’s not usually on your body.
Go to the emergency room right away if you’ve experienced trauma or think you have a fracture.
Osteoporosis
Osteoporosis weakens bones, making them more susceptible to sudden and unexpected fractures. It usually doesn’t cause any symptoms you can notice, so many people don’t know they have osteoporosis until after it causes them to break a bone.
Females and adults older than 65 have an increased risk for developing osteoporosis. Talk to your provider about a bone density test that can catch osteoporosis before it causes a fracture.
What tests are done on the ulna?
The most common test done to check the health of your ulna is a bone density test. It’s sometimes called a DEXA or DXA scan. A bone density test uses low levels of X-rays to measure how strong your bones are. It’s a way for your provider to track bone loss as you age.
If you’ve experienced a humeral fracture, your provider or surgeon might need imaging tests, including:
* X-rays.
* Magnetic Resonance Imaging (MRI).
* CT scan.
What are common treatments for the ulna?
Usually, your ulna won’t need treatment unless you’ve experienced a fracture or other injury to your arm. You might need treatment if you’ve been diagnosed with osteoporosis.
Ulna fracture treatment
How your fracture is treated depends on which type it is and what caused it. You’ll need some form of immobilization like a splint or cast. You might need surgery to realign (set) your bone to its correct position so it can heal.
Osteoporosis treatment
Treatments for osteoporosis can include exercise, vitamin and mineral supplements and medications.
Your provider will help you develop a treatment plan that’s customized for you and your bone health.

#55 Re: Jai Ganesh's Puzzles » General Quiz » 2026-03-07 16:24:47
Hi,
#10787. What does the term in Biology Histology mean?
#10788. What does the term in Biology Hodgkin–Huxley model mean?
#56 Re: Jai Ganesh's Puzzles » English language puzzles » 2026-03-07 16:12:08
Hi,
#5983. What does the verb (used with object) gall mean?
#5984. What does the noun galleon mean?
#57 Re: Jai Ganesh's Puzzles » Doc, Doc! » 2026-03-07 15:56:26
Hi,
#2588. What does the medical term Peristalsis mean?
#58 Re: Jai Ganesh's Puzzles » 10 second questions » 2026-03-07 15:40:41
Hi,
#9874.
#59 Re: Jai Ganesh's Puzzles » Oral puzzles » 2026-03-07 15:27:56
Hi,
#6367.
#60 Re: Exercises » Compute the solution: » 2026-03-07 15:09:57
Hi,
2728.
#61 Re: This is Cool » Miscellany » 2026-03-07 00:03:52
2514) Arctic Wolf
Gist:
Do Arctic wolves still exist?
They were listed as vulnerable from 1982 to 1995, but their status was upgraded to least concern in 1996 and has remained the same ever since. Thanks to their remote, icy habitat, Arctic wolves live far away from any human populations.
Are Arctic wolves friendly to humans?
The Arctic wolf is relatively unafraid of people, and can be coaxed to approach people in some areas.
Summary
The Arctic fox (Vulpes lagopus), also known as the white fox, polar fox, or snow fox, is a small species of fox native to the Arctic regions of the Northern Hemisphere and common throughout the Arctic tundra. It is well adapted to living in cold environments, and is known for its thick, warm fur that can be used as camouflage against snow in the winter. It has a large and fluffy tail. In the wild, most individuals do not live past their first year but some exceptional ones survive up to 11 years. Its body length ranges from 46 to 68 cm (18 to 27 in), with a generally rounded body shape to minimize the escape of body heat.
The Arctic fox preys on many small creatures such as lemmings, voles, ringed seal pups, fish, waterfowl, and seabirds. It also eats carrion, berries, seaweed, and insects and other small invertebrates. Arctic foxes form monogamous pairs during the breeding season and they stay together to raise their young in complex underground dens. Occasionally, other family members may assist in raising their young. Natural predators of the Arctic fox include golden eagles, Arctic wolves, polar bears, wolverines, red foxes, and brown bears.
Details
The Arctic wolf (Canis lupus arctos), also known as the white wolf, polar wolf, and the Arctic grey wolf, is a subspecies of grey wolf native to the High Arctic tundra of Canada's Queen Elizabeth Islands, from Melville Island to Ellesmere Island. Unlike some populations that move between tundra and forest regions, Arctic wolves spend their entire lives north of the northern treeline. Their southward distribution is limited to the northern fringes of the Middle Arctic tundra on the southern half of Prince of Wales and Somerset Islands.
It is a medium-sized subspecies, distinguished from the northwestern wolf by its smaller size, whiter colouration, narrower braincase, and larger carnassials. Since 1930, there has been a progressive reduction in size in Arctic wolf skulls, which is likely the result of wolf-dog hybridization.
Taxonomy
In 1935, the British zoologist Reginald Pocock attributed the subspecies name Canis lupus arctos (Arctic wolf) to a specimen from Melville Island in the Queen Elizabeth Islands, Canada. He wrote that similar wolves could be found on Ellesmere Island. He also attributed the name Canis lupus orion to a Greenland wolf specimen from Cape York, northwest Greenland. Both wolves are recognized as separate subspecies of Canis lupus in the taxonomic authority Mammal Species of the World (2005).
A study by Chambers et al. (2012) using autosomal microsatellite DNA and Mitochondrial DNA data indicate that the Arctic wolf has no unique haplotypes which suggests that its colonization of the Arctic Archipelago from the North American mainland was relatively recent, and thus not sufficient to warrant subspecies status. During a meeting assembled in 2014 by the National Center for Ecological Analysis and Synthesis of the United States Fish and Wildlife Service, one speaker, Robert K. Wayne, mentioned he disagreed with the conclusion that a subspecies had to be genetically distinct, believing that different subspecies could slowly grade into each other - suggesting that although it was impossible to determine if an individual wolf was one subspecies or the next using DNA, the population of Arctic wolves as a whole could be distinguished by the looking at the proportions of single-nucleotide polymorphisms (SNP): i.e. Arctic wolves could be distinguished by having three wolves in the putative population with a specific SNP, whereas another subspecies could be distinguished by having 20 wolves with that SNP. Wayne furthermore stated that he believed the habitat in which the wolf happened to be found was a good enough characteristic to distinguish a subspecies.
Behaviour
The Arctic wolf is relatively unafraid of people, and can be coaxed to approach people in some areas. The wolves on Ellesmere Island do not fear humans, which is thought to be due to them seeing humans so little, and they will approach humans cautiously and curiously. Otto Sverdrup wrote that during the Fram expedition, a pair of wolves shadowed one of his teammates, who kept them at a distance by waving his ski pole. In 1977, a pair of scientists were approached by six wolves on Ellesmere Island, with one animal leaping at one of the scientists and grazing a cheek. A number of incidents involving aggressive wolves have occurred in Alert, Nunavut, where the wolves have lived in close proximity to the local weather station for decades and became habituated to humans. One of these wolves attacked 3 people, was shot, and tested positive for rabies.
Very little is known about the movement of the Arctic wolves, mainly due to climate. The only time at which the wolf migrates is during the wintertime when there is complete darkness for 24 hours. This makes Arctic wolf movement hard to research. About 2,250 km (1,400 mi) south of the High Arctic, a wolf movement study took place in the wintertime in complete darkness, when the temperature was as low as −53 °C (−63 °F). The researchers found that wolves prey mainly on the muskoxen. There is no available information of the wolves' movements where the muskoxen were.
Diet
In the wild, Arctic wolves primarily prey on muskoxen and Arctic hares. They have also been found to prey on lemmings, caribou, Arctic foxes, birds, and beetles. It has been also found that Arctic wolves scavenge through garbage. This sort of food source will not always be found in the Arctic wolf's diet because of regional and seasonal availability. There is some debate whether the muskox or the Arctic hare is the primary prey for the hare-wolf-muskox predator-prey system. Evidence suggests that muskoxen provide long-term viability and other ungulates do not appear in the wolf's diet. Evidence suggesting that Arctic wolves depend more on hares claims that the mature wolf population paralleled the increase of hares rather than muskoxen availability. However, the degree of reliance between the two sources of food is uncertain and the amount of consumption between the two species also depends on the season and year. Debate continues when seasonal variations and the diet of young wolves is discussed. According to one study, muskox calves serve as a primary food source because the needs of pups are greater but another study suggests that "when hares were much more plentiful (Mech, 2000), wolves commonly fed them to their pups during summer." These differences may be attributed to location as well. Polar bears are rarely encountered by wolves, though there are two records of wolf packs killing polar bear cubs.
Conservation
The Arctic wolf is least concern, but it does face threats. In 1997, there was a decline in the Arctic wolf population and its prey, muskoxen (Ovibos moschatus), and Arctic hares (Lepus arcticus). This was due to unfavourable weather conditions during the summers for four years. Arctic wolf populations recovered the next summer when weather conditions returned to normal.
Additional Information
Arctic wolves have white fur year-round which allows them to blend into their snowy surroundings. Their coat is long and silky with soft, thick under fur. This is shed in the spring and the coat becomes shorter and less dense. The lengthy tail is bushy, and the legs are long giving it a lanky appearance. The feet are large and digitate with non-retractable claws. The forefeet have five toes and the hind feet have four. The skull is broad and the face and ears are well defined. Ears are slightly rounded and the face is less pointed than other species of wolf. This wolf is a subspecies of the gray wolf (Canis lupus). The year-round white coats and slightly shorter ears and noses distinguish them from the other subspecies of Canis lupus. They are also slightly smaller in stature. Male Arctic wolves generally weigh between 34 – 46 kg, and females between 36 – 38 kg.
Distribution
This sub-species lives primarily in the Arctic, the region located above 67 degrees north latitude. This is the area along the northern edge of the North American continent and northward to the North Pole, as well as along the eastern and northern shores of Greenland.
Habitat
The land in the Arctic is covered with snow and ice for most of the year except for brief periods during the summer months. Due to scarcity of grazing plants and resulting low density of prey species, wolves roam over large areas hunting for food.
Diet
They are predatory carnivores. They hunt in packs for caribou and musk-oxen. They also consume Arctic hares, ptarmigan, lemmings, and other small animals including nesting birds.
Reproduction
These wolves live in groups of seven to ten individuals. There is a highly complex social order within wolf packs and each pack has a dominant male and female, who bond for life. Mating between the pair takes place during the breeding season of January through to March. The gestation period for the pregnant alpha female is from 53 to 61 days. Permafrost in the Arctic makes it difficult for the wolves to dig dens. Instead, their dens are often rock outcroppings, caves, or shallow depressions in the tundra soil. The mother gives birth to two or three pups in late May to early June. Litter size is smaller for Arctic wolves. Pups are born blind and deaf. They have soft, fuzzy dark hair with small, droopy ears and blunt muzzles. They are entirely dependent on their mother; she is the only one who has contact with them at this time. She in turn relies on her mate to bring her food. At about ten days the pups’ eyes open and at three weeks they can hear. After a month the pups are able to eat meat. From then on the whole pack shares the job of feeding them, bringing meat which they regurgitate for the pups. Each member of the pack will affectionately lick, nuzzle, and sniff each pup. They become caregivers - providing food, play, and protection. Pups respond with squeaks and tail wags. They nibble and lick the feeder’s muzzle to stimulate regurgitation. They leave the den after eight to ten weeks to discover the outside world. As pups they are already establishing, through play, their future roles within the pack. When the pack hunts, one adult member will remain as a puppy sitter. Pups have grown strong enough to travel at six months of age, and from that time will join the other members of the pack learning survival skills. They become sexually mature at two to three years of age.
Adaptation
Wolves communicate with each other in a variety of ways. Clear communication is a key element to the success of a cooperative pack.
Body language: They have a rich vocabulary of visual signs that communicate social rank, mood, and intentions. Subtle changes in tail and ear positions, of body and head angle, making and breaking eye contact, and facial expressions are just a few. Tail movements for example are related to various feelings: friendliness, aggression, fear, status, social tension, threat, and submission are all communicated via tail position.
Vocalization: Wolves howl for many reasons, to assemble the pack before and after hunts, to locate members of the pack over distances, to warn neighboring packs of their presence, and apparently just for the fun of it. They often howl at a rendezvous site. The wolf’s howl can be heard up to 5 km away. They also growl, snarl, whine, yip, whimper, and bark.
Signs: They use scent marking to communicate their presence and territory boundaries to other wolves. This can be either urine or feces left on rocks and snow banks along their hunting trails. Wolves have a very good sense of smell. They can detect prey 1.6 km away and can sense an animal three days after it has gone.
Wolves are very social animals and live in family packs for protection and for hunting.
Pack structure: There is an alpha pair in each pack. The alpha male is the leader. Only the alpha couple breeds and with only one litter per pack. The number of pups is low in relationship to the number of adults. Wolves have concern for other wolves in the pack, with the younger members feeding and protecting the older wolves. All adults co-operate in feeding and educating the young. Wolf pack members protect each other.
Hunting: Life in a pack is extremely important to the survival of Arctic wolves. They live in very harsh conditions. Hunting together allows them to kill larger prey including musk oxen and caribou. A successful hunt depends on the cooperative efforts of the entire pack.
Wolves demonstrate intelligence in choosing prey: they look for old, sick, or weak animals that are easier to catch. Wolves work hard for their prey; they kill only one of every ten large mammals they chase. They eat all of the catch, including the bones. They have 42 teeth. Four large canine teeth are used for hanging onto and biting through flesh. Molars at the back are specialized shearing teeth referred to as carnassials. When hunting alone a wolf catches smaller animals.
Arctic wolves are well adapted to icy conditions. White fur allows them to blend into snowy surroundings. To help reduce heat loss their ears are small and rounded, the muzzle is short, and the fur is dense. Legs are shorter than other subspecies. They have tufts of hair between the pads of their feet.
Threats to Survival
Polar bears sometimes prey on Arctic wolves. Global climate change may affect these wolves. Exploration of the natural resources such as oil and gas will no doubt have an impact.

#62 Jokes » Miscellaneous Food Jokes - III » 2026-03-07 00:03:34
- Jai Ganesh
- Replies: 0
Q: What kind of music does candy listen to?
A: Lolli-Pop.
* * *
Q: What are the 4 major food groups?
A: Pizza, Coffee, Chocolate and Malt.
* * *
Q: What kind of witch crafts food?
A: A sand-witch.
* * *
Q: How does butter play baseball?
A: It butters up.
* * *
Q: Why do they call it Fast Food?
A: Because if you don't eat it really fast, you might actually taste it.
* * *
#63 Re: Dark Discussions at Cafe Infinity » crème de la crème » 2026-03-07 00:03:06
2451) John Franklin Enders
Gist:
Work
Many infectious diseases are caused by viruses—very small biological particles. A virus lacks metabolism of its own and cannot multiply without infecting a living cell. For a long time the prevailing opinion was that viruses could not be cultured in a laboratory. However, in 1949 John Enders, Frederick Robbins, and Thomas Weller succeeded in culturing the virus that causes polio in human muscle and tissue in a laboratory setting. This became an important step on the road toward a vaccine against polio.
Summary
John Franklin Enders (born Feb. 10, 1897, West Hartford, Conn., U.S.—died Sept. 8, 1985, Waterford, Conn.) was an American virologist and microbiologist who, with Frederick C. Robbins and Thomas H. Weller, was awarded the Nobel Prize for Physiology or Medicine for 1954 for his part in cultivating the poliomyelitis virus in nonnervous-tissue cultures, a preliminary step to the development of the polio vaccine.
Enders was a student of English literature at Harvard University (M.A., 1922) before he turned to bacterial studies there (Ph.D., 1930). His early researches contributed new and basic knowledge to problems of tuberculosis, pneumococcal infections, and resistance to bacterial diseases. In 1929 he joined the Harvard faculty as an assistant in the department of bacteriology and immunology, later advancing to assistant professor (1935) and associate professor (1942) in the university’s medical school.
In World War II he was a civilian consultant on infectious diseases to the U.S. War Department. From 1945 to 1949 he served the U.S. Army in a like capacity, with particular work on the mumps virus and rickettsial diseases. During this period Enders, with his coworkers Weller and Robbins, began research into new methods of producing in quantity the virus of poliomyelitis. Until that time the only effective method of growing the virus had been in the nerve tissue of living monkeys, and the vaccine thus produced had been proved dangerous to humans. The Enders–Weller–Robbins method of production, achieved in test tubes using cultures of nonnerve tissue from human embryos and monkeys, led to the development of the Salk vaccine for polio in 1954. Similarly, their production in the late 1950s of a vaccine against the measles led to the development of a licensed vaccine in the United States in 1963. Much of Enders’ research on viruses was conducted at the Children’s Hospital in Boston, where he had established a laboratory in 1946.
Details
John Franklin Enders (February 10, 1897 – September 8, 1985) was an American biomedical scientist and Nobel Laureate. Enders has been called "The Father of Modern Vaccines."
Life and education
Enders was born in West Hartford, Connecticut on February 10, 1897. His father, John Ostrom Enders, was CEO of the Hartford National Bank and left him a fortune of $19 million upon his death. He attended the Noah Webster School in Hartford, and graduated from St. Paul's School in Concord, New Hampshire in 1915. After attending Yale University a short time, he joined the United States Army Air Corps in 1918 as a flight instructor and a lieutenant.
After returning from World War I, he graduated from Yale, where he was a member of Scroll and Key as well as Delta Kappa Epsilon. He went into real estate in 1922, and tried several careers before choosing the biomedical field with a focus on infectious diseases, gaining a PhD at Harvard in 1930. He later joined the faculty at Children's Hospital Boston.
Enders died at his summer home in Waterford, Connecticut, aged 88, on 8 September 1985. His wife died in 2000.
Biomedical career
In 1949, Enders, Thomas Huckle Weller, and Frederick Chapman Robbins reported successful in vitro culture of an animal virus—poliovirus. The three received the 1954 Nobel Prize in Physiology or Medicine "for their discovery of the ability of poliomyelitis viruses to grow in cultures of various types of tissue".
Meanwhile, Jonas Salk applied the Enders-Weller-Robbins technique to produce large quantities of poliovirus, and then developed a polio vaccine in 1952. Upon the 1954 polio vaccine field trial, whose success Salk announced on the radio, Salk became a public hero but failed to credit the many other researchers that his effort rode upon, and was somewhat shunned by America's scientific establishment.
In 1954, Enders and Thomas C. Peebles isolated measlesvirus from an 11-year-old boy, David Edmonston. Disappointed by polio vaccine's development and involvement in some cases of polio and death—what Enders attributed to Salk's technique—Enders began development of measles vaccine. In October 1960, an Enders team began trials on 1,500 intellectually disabled children in New York City and on 4,000 children in Nigeria. Refusing credit for merely himself when The New York Times announced the measles vaccine effective on September 17, 1961, Enders wrote to the newspaper to acknowledge the work of various colleagues and the collaborative nature of the research. In 1963, a deactivated measles vaccine and an attenuated measles vaccine were introduced by Pfizer and Merck & Co., respectively.
He continued to work in virology research till the late 1970s and retired from the laboratory at the age of 80.
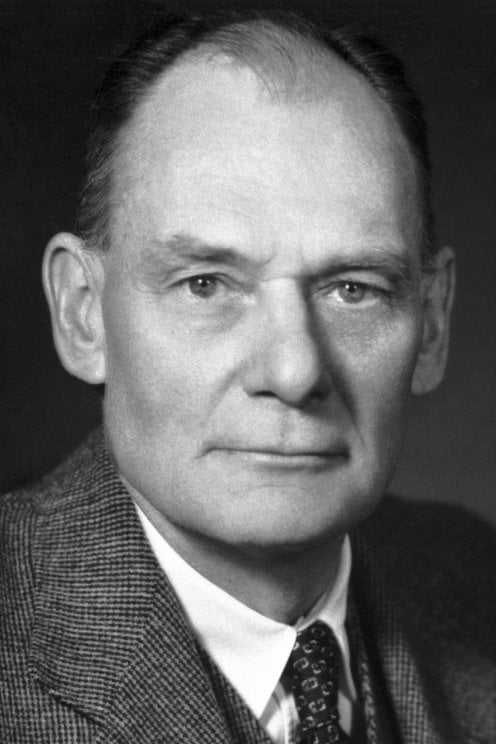
#64 Dark Discussions at Cafe Infinity » Come Quotes - XXIII » 2026-03-07 00:02:48
- Jai Ganesh
- Replies: 0
Come Quotes - XXIII
1. Logically, harmony must come from the heart... Harmony very much based on trust. As soon as use force, creates fear. Fear and trust cannot go together. - Dalai Lama
2. I perhaps ought to say that individually I never was much interested in the Texas question. I never could see much good to come of annexation, inasmuch as they were already a free republican people on our own model. - Abraham Lincoln
3. Walk while ye have the light, lest darkness come upon you. - John Ruskin
4. This is my 20th year in the sport. I've known swimming and that's it. I don't want to swim past age 30; if I continue after this Olympics, and come back in 2016, I'll be 31. I'm looking forward to being able to see the other side of the fence. - Michael Phelps
5. We don't get to know people when they come to us; we must go to them to find out what they are like. - Johann Wolfgang von Goethe
6. There are plenty of recommendations on how to get out of trouble cheaply and fast. Most of them come down to this: Deny your responsibility. - Lyndon B. Johnson
7. We all keep dreaming, and luckily, dreams come true. - Katie Holmes
8. Why are we here? Where do we come from? Traditionally, these are questions for philosophy, but philosophy is dead. - Stephen Hawking
9. The nuclear approach I'm involved in is called a traveling-wave reactor, which uses waste uranium for fuel. There's a lot of things that have to go right for that dream to come true - many decades of building demo plants, proving the economics are right. But if it does, you could have cheaper energy with no CO2 emissions. - Bill Gates
10. I really don't like going out anymore. I used to love it, but now it's not fun. I'd rather have friends come over and hot have to worry about crazy people taking pictures. - Paris Hilton
11. There were times I wouldn't come to practice, because it didn't excite me. It wasn't interesting. I was kind of going through the motions. - Michael Phelps.
#65 This is Cool » River Congo - The Deepest River In The World » 2026-03-06 18:10:53
- Jai Ganesh
- Replies: 0
River Congo - The Deepest River In The World
Gist
The Congo River is Africa's second-longest (4,700 km) and the world's deepest river (>220 m), flowing in a massive arc through Central Africa to the Atlantic Ocean. It crosses the equator twice, drains the world's second-largest rainforest, and features enormous discharge volume. Key features include the Inga Falls, major biodiversity, and essential, yet challenging, navigation for transport.
The Congo River can tick a lot of impressive feats off its list: it is Africa's most powerful river, the second most voluminous river in the world, the fifth longest, and the deepest. It even passes through the second-largest rainforest in the world.
Summary
Congo River is a river in west-central Africa. With a length of 2,900 miles (4,700 km), it is the continent’s second longest river, after the Nile. It rises in the highlands of northeastern Zambia between Lakes Tanganyika and Nyasa (Malawi) as the Chambeshi River at an elevation of 5,760 feet (1,760 metres) above sea level and at a distance of about 430 miles (700 km) from the Indian Ocean. Its course then takes the form of a giant counterclockwise arc, flowing to the northwest, west, and southwest before draining into the Atlantic Ocean at Banana (Banane) in the Democratic Republic of the Congo. Its drainage basin, covering an area of 1,335,000 square miles (3,457,000 square km), takes in almost the entire territory of that country, as well as most of the Republic of the Congo, the Central African Republic, eastern Zambia, and northern Angola and parts of Cameroon and Tanzania.
With its many tributaries, the Congo forms the continent’s largest network of navigable waterways. Navigability, however, is limited by an insurmountable obstacle: a series of 32 cataracts over the river’s lower course, including the famous Inga Falls. These cataracts render the Congo unnavigable between the seaport of Matadi, at the head of the Congo estuary, and Malebo Pool, a lakelike expansion of the river. It was on opposite banks of Malebo Pool—which represents the point of departure of inland navigation—that the capitals of the former states of the French Congo and the Belgian Congo were founded: on the left bank Kinshasa (formerly Léopoldville), now the capital of the Democratic Republic of the Congo, and on the right bank Brazzaville, now the capital of the Republic of the Congo.
The Amazon and the Congo are the two great rivers of the world that flow out of equatorial zones where heavy rainfall occurs throughout all or almost all of the year. Upstream from Malebo Pool, the Congo basin receives an average of about 60 inches (1,500 mm) of rain a year, of which more than one-fourth is discharged into the Atlantic. The drainage basin of the Congo is, however, only about half the size of that of the Amazon, and the Congo’s rate of flow—1,450,000 cubic feet (41,000 cubic metres) per second at its mouth—is considerably less than the Amazon’s flow of more than 6,180,000 cubic feet (175,000 cubic metres) per second.
While the Chambeshi River, as the remotest source, may form the Congo’s original main stream in terms of the river’s length, it is another tributary—the Lualaba, which rises near Musofi in southeastern Democratic Republic of the Congo—that carries the greatest quantity of water and thus may be considered as forming the Congo’s original main stream in terms of water volume.
When the river first became known to Europeans at the end of the 15th century, they called it the Zaire, a corruption of a word that is variously given as nzari, nzali, njali, nzaddi, and niadi and that simply means “river” in local African languages. It was only in the early years of the 18th century that the river was first called the “Rio Congo,” a name taken from the kingdom of Kongo that had been situated along the lower course of the river. During the period (1971–97) when the Democratic Republic of the Congo was called Zaire, the government also renamed the river the Zaire. Even during that time, however, the river continued to be known throughout the world as the Congo. To the literary-minded the river is evocative of the famous 1902 short story “Heart of Darkness” by Joseph Conrad. His book conjured up an atmosphere of foreboding, treachery, greed, and exploitation. Today, however, the Congo appears as the key to the economic development of the central African interior.
Details
The Congo River, formerly also known as the Zaire River, is the second-longest river in Africa, shorter only than the Nile, as well as the third largest river in the world by discharge volume, following the Amazon and Ganges–Brahmaputra rivers. It is the world's deepest recorded river, with measured depths of around 220 m (720 ft). The Congo–Lualaba–Luvua–Luapula–Chambeshi River system has an overall length of 4,700 km (2,900 mi), which makes it the world's ninth-longest river. The Chambeshi is a tributary of the Lualaba River, and Lualaba is the name of the Congo River upstream of Boyoma Falls, extending for 1,800 km (1,100 mi).
Measured along with the Lualaba, the main tributary, the Congo River has a total length of 4,370 km (2,720 mi). It is the only major river to cross the equator twice. The Congo Basin has a total area of about 4,000,000 sq km (1,500,000 sq mi), or 13% of the entire African landmass.
Name
The name Congo/Kongo originates from the Kingdom of Kongo once located on the southern bank of the river. The kingdom in turn was named after the indigenous Bantu Kongo people, known in the 17th century as "Esikongo". South of the Kingdom of Kongo proper lay the similarly named Kakongo kingdom, mentioned in 1535. Abraham Ortelius labelled "Manicongo" as the city at the mouth of the river in his world map of 1564. The tribal names in Kongo possibly derive from a word for a public gathering or tribal assembly. The modern name of the Kongo people or Bakongo was introduced in the early 20th century.
The name Zaire is from a Portuguese adaptation of a Kikongo word, nzere ("river"), a truncation of nzadi o nzere ("river swallowing rivers"). The river was known as Zaire during the 16th and 17th centuries; Congo seems to have replaced Zaire gradually in English usage during the 18th century, and Congo is the preferred English name in 19th-century literature, although references to Zahir or Zaire as the name used by the inhabitants remained common. The Democratic Republic of the Congo and the Republic of the Congo are named after it, as was the previous Republic of the Congo which had gained independence in 1960 from the Belgian Congo. The Republic of Zaire during 1971–1997 was also named after the river's name in French and Portuguese.
Basin and course
The Congo's drainage basin covers 4,014,500 sq km (1,550,000 sq mi),[6] an area nearly equal to that of the European Union. The Congo's discharge at its mouth ranges from 23,000 to 75,000 {m^3}/s (810,000 to 2,650,000 cu ft/s), with an average of 41,000 {m^3}/s (1,400,000 cu ft/s). The river transports annually 86 million tonnes of suspended sediment to the Atlantic Ocean and an additional 6% of bedload.
The river and its tributaries flow through the Congo rainforest, the second largest rainforest area in the world, after the Amazon rainforest in South America. The third-largest river in the world by discharge volume (at mouth 41,860 m3/s), following the Amazon (219,530 {m^3}/s) and Ganges–Brahmaputra–Meghna (total discharge at delta 43,950 {m}^3/s) rivers;[8] the second-largest drainage basin of any river, behind the Amazon; and is one of the deepest rivers in the world, at depths greater than 220 m (720 ft). Because its drainage basin includes areas both north and south of the Equator, its flow is stable, as there is always at least one part of the river experiencing a rainy season.
The sources of the Congo are in the highlands and mountains of the East African Rift, as well as Lake Tanganyika and Lake Mweru, which feed the Lualaba River, which then becomes the Congo below Boyoma Falls. The Chambeshi River in Zambia is generally taken as the source of the Congo in line with the accepted practice worldwide of using the longest tributary, as with the Nile River.
The Congo flows generally toward the northwest from Kisangani just below the Boyoma Falls, then gradually bends southwestward, passing by Mbandaka, joining with the Ubangi River and running into the Pool Malebo (Stanley Pool). Kinshasa (formerly Léopoldville) and Brazzaville are on opposite sides of the river at the Pool, where the river narrows and falls through a number of cataracts in deep canyons (collectively known as the Livingstone Falls), running by Matadi and Boma, and into the sea at Muanda.
Lower Congo constitutes the "lower" parts of the great river; that is the section of the river from the river mouth at the Atlantic coast to the twin capitals of Brazzaville and Kinshasa. In this section of the river, there are two significant tributaries, both on the left or south side. The Kwilu River originates in the hills near the Angolan border and enters the Congo some 100 km upstream from Matadi. The other is the Inkisi River, that flows in a northerly direction from the Uíge Province in Angola to the confluence with the Congo at Zongo some 80 km (50 mi) downstream from the twin capitals. Because of the vast number of rapids, in particular the Livingstone Falls, this section of the river is not operated continuously by riverboats.
Drainage basin
The Congo basin covers ten countries and accounts for about 13% of Africa. The highest point in the Congo basin is in the Ruwenzori Mountains, at an altitude of around 4,340 m (14,240 ft) above sea level.
Economic importance
Although the Livingstone Falls prevent access from the sea, nearly the entire Congo above them is readily navigable in sections, especially between Kinshasa and Kisangani. Large river steamers worked the river until quite recently. The Congo River still is a lifeline in a land with few roads or railways. Railways now bypass the three major falls, and much of the trade of Central Africa passes along the river, including copper, palm oil (as kernels), sugar, coffee, and cotton.
Hydroelectric power
The Congo River is the most powerful river in Africa. During the rainy season over 50,000 cubic metres (1,800,000 cu ft) of water per second flows into the Atlantic Ocean. Opportunities for the Congo River and its tributaries to generate hydropower are therefore enormous. Scientists have calculated that the entire Congo Basin accounts for 13 percent of global hydropower potential. This would provide sufficient power for all of Sub-Saharan Africa's electricity needs.
Currently, there are about 40 hydropower plants in the Congo Basin. The largest are the Inga dams, about 200 kilometres (120 mi) southwest of Kinshasa. The project was launched in the early 1970s, when the first dam was completed. The plan (as originally conceived) called for the construction of five dams that would have had a total generating capacity of 34,500 megawatts (MW). To date only the Inga I and Inga II dams have been built, generating 1,776 MW.
In February 2005, South Africa's state-owned power company, Eskom, announced a proposal to expand generation through improvements and the construction of a new hydroelectric dam. The project would bring the maximum output of the facility to 40,000 megawatts (MW). It is feared that these new hydroelectric dams could lead to the extinction of many of the fish species that are native to the river.
Natural history
The current course of the Congo River formed between 1.5 and 2 million years BP, during the Pleistocene. It is likely that during this period many upper tributaries of the Congo were captured from adjacent river basins, including the Uele and upper Ubangi from the Chari system and the Chambeshi River alongside a number of upper Kasai River tributaries from the Zambezi system.
The Congo's formation may have led to the allopatric speciation of the bonobo and the common chimpanzee from their most recent common ancestor. The bonobo is endemic to the humid forests in the region, as are other iconic species like the Allen's swamp monkey, dryas monkey, aquatic genet, okapi, and Congo peafowl.
In terms of aquatic life, the Congo River Basin has a very high species richness and among the highest known densities of endemics. As of 2009, almost 800 fish species have been recorded from the Congo River Basin (not counting Lake Tanganyika, which is connected but ecologically very different), and large sections remain virtually unstudied. For example, the section in Salonga National Park, which is about the size of Belgium, had still not been sampled at all in 2006. New fish species are scientifically described with some regularity from the Congo River Basin, and many undescribed species are known.
The Congo has by far the highest diversity of any African river system; in comparison, the next richest are the Niger, Volta and Nile with about 240, 140 and 130 fish species, respectively. Because of the great ecological differences between the regions in the Congo basin—including habitats such as river rapids, deep rivers, swamps, and lakes—it is often divided into multiple ecoregions (instead of treating it as a single ecoregion). Among these ecoregions, the Livingstone Falls cataracts has more than 300 fish species, including approximately 80 endemics while the southwestern part (Kasai River basin) has more than 200 fish species, of which about a quarter are endemic.
The dominant fish families—at least in parts of the river—are Cyprinidae (carp/cyprinids, such as Labeo simpsoni), Mormyridae (elephant fishes), Alestidae (African tetras), Mochokidae (squeaker catfishes), and Cichlidae (cichlids). Among the natives in the river is the huge, highly carnivorous giant tigerfish. Three of the more unusual endemics are the whitish (non-pigmented) and blind Lamprologus lethops, which is believed to live as deep as 160 metres (520 ft) below the surface, Heterochromis multidens, which is more closely related to cichlids of the Americas than other African cichlids, and Caecobarbus geertsii, the only known cavefish in Central Africa. There are also numerous endemic frogs and snails. Several hydroelectric dams are planned on the river, and these may lead to the extinction of many of the endemics.
Several species of turtles and the slender-snouted, Nile and dwarf crocodile are native to the Congo River Basin. African manatees inhabit the lower parts of the river.
Flooding
The Democratic Republic of Congo (DRC) and Republic of Congo (RoC) have historically experienced recurrent flooding along the Congo River and its main tributaries during the rainy season, causing erosion and landslides, and negatively impacting human settlements, agriculture, and public health. The Congo River is expected to be increasingly impacted by climate change as temperatures rise and rainfall increases both in intensity and seasonal duration.
From October 2019 to January 2020, heavy rains affected 16 of the 26 provinces of the DRC and eight out of 12 departments of the RoC, leading to the 2019–2020 Congo River floods. The rains caused the overflow of the Congo River and Ubangi rivers, floods and landslides throughout the DRC and RoC and led to the displacement of hundreds of thousands of people.
Additional Information
The Congo River (also known as Zaire River) is the largest river in Africa. Its overall length of 4,700 km (2,922 miles) makes it the second longest in Africa (after the Nile). The river and its tributaries flow through the second largest rain forest area in the world, second only to the Amazon Rainforest in South America.
The river also has the second-largest flow in the world, behind the Amazon, and the second-largest watershed of any river, again trailing the Amazon. Its watershed is a little larger than that of the Mississippi River. Because large parts of the river basin sit north and south of the equator, its flow is steady, as there is always at least one river having a rainy season. The Congo gets its name from the old Kingdom of Kongo which was at the mouth of the river. The Democratic Republic of the Congo and the Republic of the Congo, both countries sitting along the river's banks, are named after it. From 1971 to 1997, the Democratic Republic of the Congo was called Zaire and its government called the river the Zaire River.
The sources of the Congo are in the Highlands and mountains of the East African Rift, as well as Lake Tanganyika and Lake Mweru, which feed the Lualaba River. This then becomes the Congo below Boyoma Falls. The Chambeshi River in Zambia is usually taken as the source of the Congo because of the accepted practice worldwide of using the longest tributary, as with the Nile River.
The Congo flows mostly west from Kisangani just below the falls, then slowly bends southwest, passing by Mbandaka, joining with the Ubangi River, and running into the Pool Malebo (Stanley Pool). Kinshasa (formerly Léopoldville) and Brazzaville are on opposite sides of the river at the Pool, where the river narrows and falls through a few cataracts in deep canyons (collectively known as the Livingstone Falls), running by Matadi and Boma, and into the sea at the small town of Muanda.
History of exploration
The mouth of the Congo was visited by Europeans in 1482, by the Portuguese Diogo Cão, and in 1817, by a British exploration under James Kingston Tuckey that went up the river as far as Isangila. Henry Morton Stanley was the first European to travel along the whole river.
Economic importance
Although the Livingstone Falls stop ships coming in from the sea, almost all of the Congo is navigable in parts, especially between Kinshasa and Kisangani. Railways cross the three major falls that interrupt navigation, and much of the trade of central Africa passes along the river. Goods include copper, palm oil, sugar, coffee, and cotton. The river can also be valuable for hydroelectric power, and Inga Dams below Pool Malebo have been built.
In February of 2005, South Africa's state owned power company, Eskom, said that they had a proposal to increase the amount of electric power that the Inga can make through improvements and the building of a new hydroelectric dam. The project would bring the highest output of the dam to 40 GW, twice that of China's Three Gorges Dam.
Geological history
In the Mesozoic period before the continental drift opened the South Atlantic Ocean, the Congo was the upper part of a river about 12,000 km (7,500 miles) long that flowed west across the parts of Gondwanaland, now called Africa and South America.

#66 Science HQ » Femur » 2026-03-06 17:15:01
- Jai Ganesh
- Replies: 0
Femur
Gist
The femur, or thigh bone, is the longest, strongest, and heaviest bone in the human body, extending from the hip to the knee. It is critical for weight-bearing, walking, and running. Key structures include the ball-shaped head (hip joint), a strong shaft, and the condyles (knee joint).
The femur, or thigh bone, is the body's longest, strongest, and heaviest bone, running from the hip to the knee, supporting body weight, and enabling leg movement for activities like walking, running, and jumping. It connects to the pelvis at the hip (forming the hip joint) and the tibia (shin bone) at the knee, acting as a crucial pillar and anchor point for powerful muscles.
Summary:
What Is the Femur?
The femur is the longest, strongest, and heaviest bone in the human body, making it a difficult one to break. It’s also protected by various muscles and helps you maintain your posture and balance.
Femur bone function
Your femur connects many important muscles, tendons, and ligaments in your hips and knees to the other parts of your body, including parts of the circulatory system. More than that, it helps you carry your body weight when you stand and move. Your femur is critical in helping you maintain stability so you don’t fall over easily.
The femur also contains bone marrow, which is a soft, fatty tissue made up of stem cells. Stem cells have two unique abilities that make them essential to survival — they can make more stem cells, and they can “morph” or develop into other types of cells (this process is called “differentiation”).
These stem cells form two types of bone marrow: red and yellow. Each has an important job. Red bone marrow cells produce all the components of your blood (red and white blood cells and blood platelets). Yellow bone marrow cells store fat, which is needed for energy and to produce bone, cartilage, and muscles. From birth to around age 7, your bones contain only red marrow. From then on, yellow bone marrow gradually replaces red.
Details
The femur or thigh bone is the only bone in the thigh — the region of the lower limb between the hip and the knee. In many four-legged animals, the femur is the upper bone of the hindleg.
The top of the femur fits into a socket in the pelvis called the hip joint, and the bottom of the femur connects to the shinbone (tibia) and kneecap (patella) to form the knee. In humans the femur is the largest and thickest bone in the body.
Structure
The femur is the only bone in the upper leg and the longest bone in the human body. The two femurs converge medially toward the knees, where they articulate with the proximal ends of the tibiae. The angle at which the femora converge is an important factor in determining the femoral-tibial angle. In females, thicker pelvic bones cause the femora to converge more than in males.
In the condition genu valgum (knock knee), the femurs converge so much that the knees touch. The opposite condition, genu varum (bow-leggedness), occurs when the femurs diverge. In the general population without these conditions, the femoral-tibial angle is about 175 degrees.
The femur is the thickest bone in the human body. It is considered the strongest bone by some measures, though other studies suggest the temporal bone may be stronger. On average, the femur length accounts for 26.74% of a person's height, a ratio found in both men and women across most ethnic groups with minimal variation. This ratio is useful in anthropology, as it provides a reliable estimate of a person's height from an incomplete skeleton.
The femur is classified as a long bone, consisting of diaphysis (shaft or body) and two epiphyses (extremities) that articulate with the hip and knee bones.
Upper part
The upper or proximal extremity (close to the torso) contains the head, neck, the two trochanters and adjacent structures. The upper extremity is the thinnest femoral extremity, the lower extremity is the thickest femoral extremity.
The head of the femur, which articulates with the acetabulum of the pelvic bone, comprises two-thirds of a sphere. It has a small groove, or fovea, connected through the round ligament to the sides of the acetabular notch. The head of the femur is connected to the shaft through the neck or collum. The neck is 4–5 cm. long and the diameter is smallest front to back and compressed at its middle. The collum forms an angle with the shaft in about 130 degrees. This angle is highly variant. In the infant, it is about 150 degrees and in old age reduced to 120 degrees on average. An abnormal increase in the angle is known as coxa valga and an abnormal reduction is called coxa vara. Both the head and neck of the femur is vastly embedded in the hip musculature and can not be directly palpated. In skinny people with the thigh laterally rotated, the head of the femur can be felt deep as a resistance profound (deep) for the femoral artery.
The transition area between the head and neck is quite rough due to attachment of muscles and the hip joint capsule. Here the two trochanters, greater and lesser trochanter, are found. The greater trochanter is almost box-shaped and is the most lateral prominent of the femur. The highest point of the greater trochanter is located higher than the collum and reaches the midpoint of the hip joint. The greater trochanter can easily be felt. The trochanteric fossa is a deep depression bounded posteriorly by the intertrochanteric crest on the medial surface of the greater trochanter. The lesser trochanter is a cone-shaped extension of the lowest part of the femur neck. The two trochanters are joined by the intertrochanteric crest on the back side and by the intertrochanteric line on the front.
A slight ridge is sometimes seen commencing about the middle of the intertrochanteric crest, and reaching vertically downward for about 5 cm. along the back part of the body: it is called the linea quadrata (or quadrate line).
About the junction of the upper one-third and lower two-thirds on the intertrochanteric crest is the quadrate tubercle located. The size of the tubercle varies and it is not always located on the intertrochanteric crest and that also adjacent areas can be part of the quadrate tubercle, such as the posterior surface of the greater trochanter or the neck of the femur. In a small anatomical study it was shown that the epiphyseal line passes directly through the quadrate tubercle.
Body
The body of the femur (or shaft) is large, thick and almost cylindrical in form. It is a little broader above than in the center, broadest and somewhat flattened from before backward below. It is slightly arched, so as to be convex in front, and concave behind, where it is strengthened by a prominent longitudinal ridge, the linea aspera which diverges proximally and distal as the medial and lateral ridge. Proximally the lateral ridge of the linea aspera becomes the gluteal tuberosity while the medial ridge continues as the pectineal line. Besides the linea aspera the shaft has two other bordes; a lateral and medial border. These three bordes separate the shaft into three surfaces: One anterior, one medial and one lateral. Due to the vast musculature of the thigh the shaft can not be palpated.
The third trochanter is a bony projection occasionally present on the proximal femur near the superior border of the gluteal tuberosity. When present, it is oblong, rounded, or conical in shape and sometimes continuous with the gluteal ridge. A structure of minor importance in humans, the incidence of the third trochanter varies from 17–72% between ethnic groups and it is frequently reported as more common in females than in males.
Lower part
The lower extremity of the femur (or distal extremity) is the thickest femoral extremity, the upper extremity is the shortest femoral extremity. It is somewhat cuboid in form, but its transverse diameter is greater than its antero-posterior (front to back). It consists of two oblong eminences known as the condyles.
Anteriorly, the condyles are slightly prominent and are separated by a smooth shallow articular depression called the patellar surface. Posteriorly, they project considerably and a deep notch, the Intercondylar fossa of femur, is present between them. The lateral condyle is the more prominent and is the broader both in its antero-posterior and transverse diameters. The medial condyle is the longer and, when the femur is held with its body perpendicular, projects to a lower level. When, however, the femur is in its natural oblique position the lower surfaces of the two condyles lie practically in the same horizontal plane. The condyles are not quite parallel with one another; the long axis of the lateral is almost directly antero-posterior, but that of the medial runs backward and medialward. Their opposed surfaces are small, rough, and concave, and form the walls of the intercondyloid fossa. This fossa is limited above by a ridge, the intercondyloid line, and below by the central part of the posterior margin of the patellar surface. The posterior cruciate ligament of the knee joint is attached to the lower and front part of the medial wall of the fossa and the anterior cruciate ligament to an impression on the upper and back part of its lateral wall.
The articular surface of the lower end of the femur occupies the anterior, inferior, and posterior surfaces of the condyles. Its front part is named the patellar surface and articulates with the patella; it presents a median groove which extends downward to the intercondyloid fossa and two convexities, the lateral of which is broader, more prominent, and extends farther upward than the medial.
Each condyle is surmounted by an elevation, the epicondyle. The medial epicondyle is a large convex eminence to which the tibial collateral ligament of the knee-joint is attached. At its upper part is the adductor tubercle and behind it is a rough impression which gives origin to the medial head of the gastrocnemius. The lateral epicondyle which is smaller and less prominent than the medial, gives attachment to the fibular collateral ligament of the knee-joint.
Development
The femur develops from the limb buds as a result of interactions between the ectoderm and the underlying mesoderm; formation occurs roughly around the fourth week of development.
By the sixth week of development, the first hyaline cartilage model of the femur is formed by chondrocytes. Endochondral ossification begins by the end of the embryonic period and primary ossification centers are present in all long bones of the limbs, including the femur, by the 12th week of development. The hindlimb development lags behind forelimb development by 1–2 days.
Function
As the femur is the only bone in the thigh, it serves as an attachment point for all the muscles that exert their force over the hip and knee joints. Some biarticular muscles – which cross two joints, like the gastrocnemius and plantaris muscles – also originate from the femur. In all, 23 individual muscles either originate from or insert onto the femur.
In cross-section, the thigh is divided up into three separate fascial compartments divided by fascia, each containing muscles. These compartments use the femur as an axis, and are separated by tough connective tissue membranes (or septa). Each of these compartments has its own blood and nerve supply, and contains a different group of muscles. These compartments are named the anterior, medial and posterior fascial compartments.
Clinical significance:
Fractures
A femoral fracture that involves the femoral head, femoral neck or the shaft of the femur immediately below the lesser trochanter may be classified as a hip fracture, especially when associated with osteoporosis. Femur fractures can be managed in a pre-hospital setting with the use of a traction splint.
Cortical desmoid
Cortical desmoid (also known as a tug lesion or periosteal desmoid) is an irregularity of the distal femoral cortex commonly observed is adolescents.
Additional Information
The femur is the longest, strongest bone in your body. It plays an important role in how you stand, move and keep your balance. Femurs usually only break from serious traumas like car accidents. But if your bones are weakened by osteoporosis, you have an increased risk for fractures you might not even know about.
Overview:
What is the femur?
The femur is your thigh bone. It’s the longest, strongest bone in your body. It’s a critical part of your ability to stand and move. Your femur also supports lots of important muscles, tendons, ligaments and parts of your circulatory system.
Because it’s so strong, it usually takes a severe trauma like a fall or car accident to break your femur. If you do experience a fracture, you’ll likely need surgery to repair your bone and physical therapy to help you regain your strength and ability to move.
Your femur, like all bones, can be affected by osteoporosis.
Function:
What does the femur do?
Your femur has several important jobs, including:
* Holding the weight of your body when you stand and move
* Stabilizing you as you move
* Connecting muscles, tendons and ligaments in your hips and knees to the rest of your body
Anatomy:
Where is the femur located?
The femur is the only bone in your thigh. It runs from your hip to your knee.
What does the femur look like?
The femur has two rounded ends and a long shaft in the middle. It’s the classic shape used for bones in cartoons: A cylinder with two round bumps at each end.
Even though it’s one long bone, your femur is made up of several parts. These include:
Femur proximal aspect
The upper (proximal) end of your femur connects to your hip joint. The proximal end (aspect) contains the:
* Head
* Neck
* Greater trochanter
* Lesser trochanter
* Intertrochanteric line and crest
Femur shaft
The shaft is the long portion of the femur that supports your weight and forms the structure of your thigh. It angles slightly toward the center of your body. The shaft of your femur includes the:
* Linea aspera
* Gluteal tuberosity
* Pectineal line
* Popliteal fossa
Femur distal aspect
The lower (distal) end of your femur forms the top of your knee joint. It meets your tibia (shin) and patella (kneecap). It includes the:
* Medial and lateral condyles
* Medial and lateral epicondyles
* Intercondylar fossa
All of these parts and labels are usually more for your healthcare provider to use as they describe where you’re having pain or issues. If you ever break your femur — a femoral fracture — your provider might use some of these terms to describe where your bone was damaged.
How big is the femur?
Your femur is the largest bone in your body. Most adult femurs are around 18 inches long.
The femur is also the strongest bone in your body. It can support as much as 30 times the weight of your body.
Conditions and Disorders:
What are the common conditions and disorders that affect the femur?
The most common issues that affect femurs are fractures, osteoporosis and patellofemoral pain syndrome.
Femur fractures
A bone fracture is the medical term for breaking a bone. Because femurs are so strong, they’re usually only broken by serious injuries like car accidents, falls or other traumas. Symptoms of a fracture include:
* Pain
* Swelling
* Tenderness
* Inability to move your leg like you usually can
* Bruising or discoloration
* A deformity or bump that’s not usually on your body
Go to the emergency room right away if you’ve experienced a trauma or think you have a fracture.
Osteoporosis
Osteoporosis weakens bones, making them more susceptible to sudden and unexpected fractures. Many people don’t know they have osteoporosis until after it causes them to break a bone. There usually aren’t obvious symptoms.
Females and adults older than 50 have an increased risk of developing osteoporosis. Talk to your provider about a bone density screening that can catch osteoporosis before it causes a fracture.
Patellofemoral pain syndrome
Patellofemoral pain syndrome (PFPS) is pain around and under your kneecap (patella). It’s sometimes called runner’s or jumper’s knee. PFPS can be caused by everything from overusing your knees to getting new shoes. Symptoms of PFPS include:
* Pain while bending your knee, including squatting or climbing stairs
* Pain after sitting with your knees bent
* Crackling or popping sounds in your knee when standing up or climbing stairs
* Pain that increases with changes to your usual playing surface, sports equipment or activity intensity
Talk to your provider if you’re experiencing new pain in your knee.
What tests are done on femurs?
The most common test done to check the health of your femur is a bone density test. It’s sometimes called a DEXA or DXA scan. A bone density test measures how strong your bones are with low levels of X-rays. It’s a way to measure bone loss as you age.
If you’ve experienced a femoral fracture your provider or surgeon might need imaging tests, including:
* X-rays
* Magnetic resonance imaging (MRI)
* CT scan
What are common treatments for femurs?
Usually, your femur won’t need treatment unless you’ve experienced a fracture or have been diagnosed with osteoporosis.
Femur fracture treatment
How your fracture is treated depends on which type it is and what caused it. You’ll need some form of immobilization, like a splint or cast, and will probably need surgery to realign (set) your bone to its correct position and secure it in place so it can heal.
Osteoporosis treatment
Treatments for osteoporosis can include exercise, vitamin and mineral supplements and medications.
Exercise and taking supplements are usually all you’ll need to prevent osteoporosis. Your provider will help you develop a treatment plan that’s customized for you and your bone health.

#67 Re: Jai Ganesh's Puzzles » General Quiz » 2026-03-06 16:17:09
Hi,
#10785. What does the term in Geography Demonym mean?
#10786. What does the term in Geography Denudation mean?
#68 Re: Jai Ganesh's Puzzles » English language puzzles » 2026-03-06 15:38:22
Hi,
#5981. What does the noun layout mean?
#5982. What does the verb (used with object) leach mean?
#69 Re: Jai Ganesh's Puzzles » Doc, Doc! » 2026-03-06 15:25:28
Hi,
#2587. What does the medical term Coombs test signify?
#70 Re: Jai Ganesh's Puzzles » 10 second questions » 2026-03-06 14:46:28
Hi,
#9873.
#71 Re: Jai Ganesh's Puzzles » Oral puzzles » 2026-03-06 14:36:11
Hi,
#6366.
#72 Re: Exercises » Compute the solution: » 2026-03-06 14:20:47
Hi,
2727.
#73 Re: Dark Discussions at Cafe Infinity » crème de la crème » 2026-03-06 00:05:02
2450) Linus Pauling
Gist:
Life
Linus Pauling was born in Portland, Oregon, in the United States. His family came from a line of Prussian farmers, and Pauling's father worked as a pharmaceuticals salesman, among other things. After first studying at Oregon State University in Corvallis, Oregon, Pauling earned his PhD from the California Institute of Technology in Pasadena, with which he maintained ties for the rest of his career. In the 1950s, Pauling's involvement in the anti-nuclear movement led to his being labeled a suspected communist, which resulted in his passport being revoked at times. Linus and Ava Helen Pauling had four children together.
Work
The development of quantum mechanics during the 1920s had a great impact not only on the field of physics, but also on chemistry. During the 1930s Linus Pauling was among the pioneers who used quantum mechanics to understand and describe chemical bonding–that is, the way atoms join together to form molecules. Pauling worked in a broad range of areas within chemistry. For example, he worked on the structures of biologically important chemical compounds. In 1951 he published the structure of the alpha helix, which is an important basic component of many proteins.
Summary
Linus Carl Pauling (February 28, 1901 – August 19, 1994) was an American chemist and peace activist. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. New Scientist called him one of the 20 greatest scientists of all time. For his scientific work, Pauling was awarded the Nobel Prize in Chemistry in 1954. For his peace activism, he was awarded the Nobel Peace Prize in 1962. He is one of five people to have won more than one Nobel Prize. Of these, he is the only person to have been awarded two unshared Nobel Prizes, and one of two people to be awarded Nobel Prizes in different fields, the other being Marie Skłodowska-Curie.
Pauling was one of the founders of the fields of quantum chemistry and molecular biology. His contributions to the theory of the chemical bond include the concept of orbital hybridisation and the first accurate scale of electronegativities of the elements. Pauling also worked on the structures of biological molecules, and showed the importance of the alpha helix and beta sheet in protein secondary structure. Pauling's approach combined methods and results from X-ray crystallography, molecular model building, and quantum chemistry. His discoveries inspired the work of James Watson, Francis Crick, Rosalind Franklin, and Maurice Wilkins on the structure of DNA, which in turn made it possible for geneticists to crack the DNA code of all organisms.
In his later years, he promoted nuclear disarmament, as well as orthomolecular medicine, megavitamin therapy, and dietary supplements, especially ascorbic acid (commonly known as Vitamin C). None of his ideas concerning the medical usefulness of large doses of vitamins have gained much acceptance in the mainstream scientific community. He was married to the American human rights activist Ava Helen Pauling.
Details
Linus Pauling (born February 28, 1901, Portland, Oregon, U.S.—died August 19, 1994, Big Sur, California) was an American theoretical physical chemist who became the only person to have won two unshared Nobel Prizes. His first prize (1954) was awarded for research into the nature of the chemical bond and its use in elucidating molecular structure; the second (1962) recognized his efforts to ban the testing of nuclear weapons.
Early life and education
Pauling was the first of three children and the only son of Herman Pauling, a pharmacist, and Lucy Isabelle (Darling) Pauling, a pharmacist’s daughter. After his early education in Condon and Portland, Oregon, he attended Oregon Agricultural College (now Oregon State University), where he met Ava Helen Miller, who would later become his wife, and where he received his Bachelor of Science degree in chemical engineering summa cum laude in 1922. He then attended the California Institute of Technology (Caltech), where Roscoe G. Dickinson showed him how to determine the structures of crystals using X rays. He received his Ph.D. in 1925 for a dissertation derived from his crystal-structure papers. Following a brief period as a National Research Fellow, he received a Guggenheim Fellowship to study quantum mechanics in Europe. He spent most of the 18 months at Arnold Sommerfeld’s Institute for Theoretical Physics in Munich, Germany.
Elucidation of molecular structures
After completing postdoctoral studies, Pauling returned to Caltech in 1927. There he began a long career of teaching and research. Analyzing chemical structure became the central theme of his scientific work. By using the technique of X-ray diffraction, he determined the three-dimensional arrangement of atoms in several important silicate and sulfide minerals. In 1930, during a trip to Germany, Pauling learned about electron diffraction, and upon his return to California he used this technique of scattering electrons from the nuclei of molecules to determine the structures of some important substances. This structural knowledge assisted him in developing an electronegativity scale in which he assigned a number representing a particular atom’s power of attracting electrons in a covalent bond.
To complement the experimental tool that X-ray analysis provided for exploring molecular structure, Pauling turned to quantum mechanics as a theoretical tool. For example, he used quantum mechanics to determine the equivalent strength in each of the four bonds surrounding the carbon atom. He developed a valence bond theory in which he proposed that a molecule could be described by an intermediate structure that was a resonance combination (or hybrid) of other structures. His book The Nature of the Chemical Bond, and the Structure of Molecules and Crystals (1939) provided a unified summary of his vision of structural chemistry.
The arrival of the geneticist Thomas Hunt Morgan at Caltech in the late 1920s stimulated Pauling’s interest in biological molecules, and by the mid-1930s he was performing successful magnetic studies on the protein hemoglobin. He developed further interests in protein and, together with biochemist Alfred Mirsky, Pauling published a paper in 1936 on general protein structure. In this work the authors explained that protein molecules naturally coiled into specific configurations but became “denatured” (uncoiled) and assumed some random form once certain weak bonds were broken.
On one of his trips to visit Mirsky in New York, Pauling met Karl Landsteiner, the discoverer of blood types, who became his guide into the field of immunochemistry. Pauling was fascinated by the specificity of antibody-antigen reactions, and he later developed a theory that accounted for this specificity through a unique folding of the antibody’s polypeptide chain. World War II interrupted this theoretical work, and Pauling’s focus shifted to more practical problems, including the preparation of an artificial substitute for blood serum useful to wounded soldiers and an oxygen detector useful in submarines and airplanes. J. Robert Oppenheimer asked Pauling to head the chemistry section of the Manhattan Project, but his suffering from glomerulonephritis (inflammation of the glomerular region of the kidney) prevented him from accepting this offer. For his outstanding services during the war, Pauling was later awarded the Presidential Medal for Merit.
While collaborating on a report about postwar American science, Pauling became interested in the study of sickle-cell anemia. He perceived that the sickling of cells noted in this disease might be caused by a genetic mutation in the globin portion of the blood cell’s hemoglobin. In 1949 he and his coworkers published a paper identifying the particular defect in hemoglobin’s structure that was responsible for sickle-cell anemia, which thereby made this disorder the first “molecular disease” to be discovered. At that time, Pauling’s article on the periodic law appeared in the 14th edition of Encyclopædia Britannica.
While serving as a visiting professor at the University of Oxford in 1948, Pauling returned to a problem that had intrigued him in the late 1930s—the three-dimensional structure of proteins. By folding a paper on which he had drawn a chain of linked amino acids, he discovered a cylindrical coil-like configuration, later called the alpha helix. The most significant aspect of Pauling’s structure was its determination of the number of amino acids per turn of the helix. During this same period he became interested in deoxyribonucleic acid (DNA), and early in 1953 he and protein crystallographer Robert Corey published their version of DNA’s structure, three strands twisted around each other in ropelike fashion. Shortly thereafter James Watson and Francis Crick published DNA’s correct structure, a double helix. Pauling’s efforts to modify his postulated structure had been hampered by poor X-ray photographs of DNA and by his lack of understanding of this molecule’s wet and dry forms. In 1952 he failed to visit Rosalind Franklin, working in Maurice Wilkins’s laboratory at King’s College, London, and consequently did not see her X-ray pictures of DNA. Frankin’s pictures proved to be the linchpin in allowing Watson and Crick to elucidate the actual structure. Nevertheless, Pauling was awarded the 1954 Nobel Prize for Chemistry “for his research into the nature of the chemical bond and its application to the elucidation of the structure of complex substances.”
Humanitarian activities of Linus Pauling
During the 1950s Pauling and his wife became well known to the public through their crusade to stop the atmospheric testing of nuclear weapons. In 1958 they presented an appeal for a test ban to the United Nations in the form of a document signed by 9,235 scientists from 44 countries. Pauling’s sentiments were also promulgated through his book No More War! (1958), a passionate analysis of the implications of nuclear war for humanity. In 1960 he was called upon to defend his actions regarding a test ban before a congressional subcommittee. By refusing to reveal the names of those who had helped him collect signatures, he risked going to jail—a stand initially condemned but later widely admired. His work on behalf of world peace was recognized with the 1962 Nobel Prize for Peace awarded on October 10, 1963, the date that the Nuclear Test Ban Treaty went into effect.
Pauling’s Peace Prize generated such antagonism from Caltech administrators that he left the institute in 1963. He became a staff member at the Center for the Study of Democratic Institutions in Santa Barbara, California, where his humanitarian work was encouraged. Although he was able to develop a new model of the atomic nucleus while working at the Center, his desire to perform more experimental research led him to a research professorship at the University of California in San Diego in 1967. There he published a paper on orthomolecular psychiatry that explained how mental health could be achieved by manipulating substances normally present in the body. Two years later he accepted a post at Stanford University, where he worked until 1972.
Later years
While at San Diego and Stanford, Pauling’s scientific interests centred on a particular molecule—ascorbic acid (vitamin C). He examined the published reports about this vitamin and concluded that, when taken in large enough quantities (megadoses), it would help the body fight off colds and other diseases. The outcome of his research was the book Vitamin C and the Common Cold (1970), which became a best-seller. Pauling’s interest in vitamin C in particular and orthomolecular medicine in general led, in 1973, to his founding an institute that eventually bore his name—the Linus Pauling Institute of Science and Medicine. During his tenure at this institute, he became embroiled in controversies about the relative benefits and risks of ingesting megadoses of various vitamins. The controversy intensified when he advocated vitamin C’s usefulness in the treatment of cancer. Pauling and his collaborator, the Scottish physician Ewan Cameron, published their views in Cancer and Vitamin C (1979). Their ideas were subjected to experimental animal studies funded by the institute. While these studies supported their ideas, investigations at the Mayo Clinic involving human cancer patients did not corroborate Pauling’s results.
Although he continued to receive recognition for his earlier accomplishments, Pauling’s later work provoked considerable skepticism and controversy. His cluster model of the atomic nucleus was rejected by physicists, his interpretation of the newly discovered quasicrystals received little support, and his ideas on vitamin C were rejected by the medical establishment. In an effort to raise money to support his increasingly troubled institute, Pauling published How to Live Longer and Feel Better (1986), but the book failed to become the success that he and his associates had anticipated.
Both Pauling and his wife developed cancer. Ava Helen Pauling died of stomach cancer in 1981. Ten years later Pauling discovered that he had prostate cancer. Although he underwent surgery and other treatments, the cancer eventually spread to his liver. He died at his ranch on the Big Sur coast of California.

#74 Re: This is Cool » Miscellany » 2026-03-06 00:04:02
2513) Neurotransmitters
Gist
Neurotransmitters are endogenous chemical messengers that transmit signals across a synapse from one neuron to another target cell (neuron, muscle, or gland). They are vital for brain function, influencing mood, sleep, memory, and motor control. Over 50 types exist, acting as either excitatory (triggering a response) or inhibitory (inhibiting a response).
Neurotransmitters are often referred to as the body's chemical messengers. They are the molecules used by the nervous system to transmit messages between neurons, or from neurons to muscles.
Neurotransmitters are endogenous chemical messengers that transmit signals across a synapse between neurons, muscles, or gland cells, essential for regulating body functions, emotions, and thoughts. They are classified into amino acids, peptides, monoamines, purines, and gasotransmitters, with over 50 types known. Their action is either excitatory (promoting a signal) or inhibitory (stopping a signal).
Summary
A neurotransmitter is a signaling molecule secreted by a neuron to affect another cell across a synapse. The cell receiving the signal, or target cell, may be another neuron, but could also be a gland or muscle cell.
Neurotransmitters are released from synaptic vesicles into the synaptic cleft where they are able to interact with neurotransmitter receptors on the target cell. Some neurotransmitters are also stored in large dense core vesicles. The neurotransmitter's effect on the target cell is determined by the receptor it binds to. Many neurotransmitters are synthesized from simple and plentiful precursors such as amino acids, which are readily available and often require a small number of biosynthetic steps for conversion.
Neurotransmitters are essential to the function of complex neural systems. The exact number of unique neurotransmitters in humans is unknown, but more than 100 have been identified. Common neurotransmitters include glutamate, GABA, acetylcholine, glycine, dopamine and norepinephrine.
(GABA: gamma-aminobutyric acid).
Details
Neurotransmitters are your body’s chemical messengers. They carry messages from one nerve cell across a space to the next nerve, muscle or gland cell. These messages help you move your limbs, feel sensations, keep your heart beating, and take in and respond to all information your body receives from other internal parts of your body and your environment.
What are neurotransmitters?
Neurotransmitters are chemical messengers that your body can’t function without. Their job is to carry chemical signals (“messages”) from one neuron (nerve cell) to the next target cell. The next target cell can be another nerve cell, a muscle cell or a gland.
Your body has a vast network of nerves (your nervous system) that send and receive electrical signals from nerve cells and their target cells all over your body. Your nervous system controls everything from your mind to your muscles, as well as organ functions. In other words, nerves are involved in everything you do, think and feel. Your nerve cells send and receive information from all body sources. This constant feedback is essential to your body’s optimal function.
What body functions do nerves and neurotransmitters help control?
Your nervous system controls such functions as your:
* Heartbeat and blood pressure.
* Breathing.
* Muscle movements.
* Thoughts, memory, learning and feelings.
* Sleep, healing and aging.
* Stress response.
* Hormone regulation.
* Digestion, sense of hunger and thirst.
* Senses (response to what you see, hear, feel, touch and taste).
How do neurotransmitters work?
You have billions of nerve cells in your body. Nerve cells are generally made up of three parts:
* A cell body. The cell body is vital to producing neurotransmitters and maintaining the function of the nerve cell.
* An axon. The axon carries the electrical signals along the nerve cell to the axon terminal.
* An axon terminal. This is where the electrical message is changed to a chemical signal using neurotransmitters to communicate with the next group of nerve cells, muscle cells or organs.
Neurotransmitters are located in a part of the neuron called the axon terminal. They’re stored within thin-walled sacs called synaptic vesicles. Each vesicle can contain thousands of neurotransmitter molecules.
As a message or signal travels along a nerve cell, the electrical charge of the signal causes the vesicles of neurotransmitters to fuse with the nerve cell membrane at the very edge of the cell. The neurotransmitters, which now carry the message, are then released from the axon terminal into a fluid-filled space that’s between one nerve cell and the next target cell (another nerve cell, muscle cell or gland).
In this space, called the synaptic junction, the neurotransmitters carry the message across less than 40 nanometers (nm) wide (by comparison, the width of a human hair is about 75,000 nm). Each type of neurotransmitter lands on and binds to a specific receptor on the target cell (like a key that can only fit and work in its partner lock). After binding, the neurotransmitter then triggers a change or action in the target cell, like an electrical signal in another nerve cell, a muscle contraction or the release of hormones from a cell in a gland.
What action or change do neurotransmitters transmit to the target cell?
Neurotransmitters transmit one of three possible actions in their messages, depending on the specific neurotransmitter.
* Excitatory. Excitatory neurotransmitters “excite” the neuron and cause it to “fire off the message,” meaning, the message continues to be passed along to the next cell. Examples of excitatory neurotransmitters include glutamate, epinephrine and norepinephrine.
* Inhibitory. Inhibitory neurotransmitters block or prevent the chemical message from being passed along any farther. Gamma-aminobutyric acid (GABA), glycine and serotonin are examples of inhibitory neurotransmitters.
* Modulatory. Modulatory neurotransmitters influence the effects of other chemical messengers. They “tweak” or adjust how cells communicate at the synapse. They also affect a larger number of neurons at the same time.
What happens to neurotransmitters after they deliver their message?
After neurotransmitters deliver their message, the molecules must be cleared from the synaptic cleft (the space between the nerve cell and the next target cell). They do this in one of three ways.
Neurotransmitters:
* Fade away (a process called diffusion).
* Are reabsorbed and reused by the nerve cell that released it (a process called reuptake).
* Are broken down by enzymes within the synapse so it can’t be recognized or bind to the receptor cell (a process called degradation).
How many different types of neurotransmitters are there?
Scientists know of at least 100 neurotransmitters and suspect there are many others that have yet to be discovered. They can be grouped into types based on their chemical nature. Some of the better-known categories and neurotransmitter examples and their functions include the following:
Amino acids neurotransmitters
These neurotransmitters are involved in most functions of your nervous system.
* Glutamate. This is the most common excitatory neurotransmitter of your nervous system. It’s the most abundant neurotransmitter in your brain. It plays a key role in cognitive functions like thinking, learning and memory. Imbalances in glutamate levels are associated with Alzheimer’s disease, dementia, Parkinson’s disease and seizures.
* Gamma-aminobutryic acid (GABA). GABA is the most common inhibitory neurotransmitter of your nervous system, particularly in your brain. It regulates brain activity to prevent problems in the areas of anxiety, irritability, concentration, sleep, seizures and depression.
* Glycine. Glycine is the most common inhibitory neurotransmitter in your spinal cord. Glycine is involved in controlling hearing processing, pain transmission and metabolism.
Monoamines neurotransmitters
These neurotransmitters play a lot of different roles in your nervous system and especially in your brain. Monoamines neurotransmitters regulate consciousness, cognition, attention and emotion. Many disorders of your nervous system involve abnormalities of monoamine neurotransmitters, and many drugs that people commonly take affect these neurotransmitters.
* Serotonin. Serotonin is an inhibitory neurotransmitter. Serotonin helps regulate mood, sleep patterns, sexuality, anxiety, appetite and pain. Diseases associated with serotonin imbalance include seasonal affective disorder, anxiety, depression, fibromyalgia and chronic pain. Medications that regulate serotonin and treat these disorders include selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs).
* Histamine. Histamine regulates body functions including wakefulness, feeding behavior and motivation. Histamine plays a role in asthma, bronchospasm, mucosal edema and multiple sclerosis.
* Dopamine. Dopamine plays a role in your body’s reward system, which includes feeling pleasure, achieving heightened arousal and learning. Dopamine also helps with focus, concentration, memory, sleep, mood and motivation. Diseases associated with dysfunctions of the dopamine system include Parkinson’s disease, schizophrenia, bipolar disease, restless legs syndrome and attention deficit hyperactivity disorder (ADHD). Many highly addictive drugs (cocaine, methamphetamines, amphetamines) act directly on the dopamine system.
* Epinephrine. Epinephrine (also called adrenaline) and norepinephrine (see below) are responsible for your body’s so-called “fight-or-flight response” to fear and stress. These neurotransmitters stimulate your body’s response by increasing your heart rate, breathing, blood pressure, blood sugar and blood flow to your muscles, as well as heighten attention and focus to allow you to act or react to different stressors. Too much epinephrine can lead to high blood pressure, diabetes, heart disease and other health problems. As a drug, epinephrine is used to treat anaphylaxis, asthma attacks, cardiac arrest and severe infections.
* Norepinephrine. Norepinephrine (also called noradrenaline) increases blood pressure and heart rate. It’s most widely known for its effects on alertness, arousal, decision-making, attention and focus. Many medications (stimulants and depression medications) aim to increase norepinephrine levels to improve focus or concentration to treat ADHD or to modulate norepinephrine to improve depression symptoms.
Peptide neurotransmitters
Peptides are polymers or chains of amino acids.
* Endorphins. Endorphins are your body’s natural pain reliever. They play a role in our perception of pain. Release of endorphins reduces pain, as well as causes “feel good” feelings. Low levels of endorphins may play a role in fibromyalgia and some types of headaches.
Acetylcholine
This excitatory neurotransmitter does a number of functions in your central nervous system (CNS [brain and spinal cord]) and in your peripheral nervous system (nerves that branch from the CNS). Acetylcholine is released by most neurons in your autonomic nervous system regulating heart rate, blood pressure and gut motility. Acetylcholine plays a role in muscle contractions, memory, motivation, sexual desire, sleep and learning. Imbalances in acetylcholine levels are linked with health issues, including Alzheimer’s disease, seizures and muscle spasms.
Why would a neurotransmitter not work as it should?
Several things can go haywire and lead to neurotransmitters not working as they should. In general, some of these problems include:
* Too much or not enough of one or more neurotransmitters are produced or released.
* The receptor on the receiver cell (the nerve, muscle or gland) isn’t working properly. The otherwise normal functioning neurotransmitter can’t effectively signal the next cell.
* The cell receptors aren’t taking up enough neurotransmitter due to inflammation and damage of the synaptic cleft.
* Neurotransmitters are reabsorbed too quickly.
* Enzymes limit the number of neurotransmitters from reaching their target cell.
Problems with other parts of nerves, existing diseases or medications you may be taking can affect neurotransmitters. Also, when neurotransmitters don’t function as they should, disease can happen. For example:
* Not enough acetylcholine can lead to the loss of memory that’s seen in Alzheimer’s disease.
* Too much serotonin is possibly associated with autism spectrum disorders.
* An increase in activity of glutamate or reduced activity of GABA can result in sudden, high-frequency firing of local neurons in your brain, which can cause seizures.
* Too much norepinephrine and dopamine activity and abnormal glutamate transmission contribute to mania.
How do medications affect the action of neurotransmitters?
Scientists recognized the value and the role of neurotransmitters in your nervous system and the importance of developing medications that could influence these chemical messengers to treat many health conditions. Many medications, especially those that treat diseases of your brain, work in many ways to affect neurotransmitters.
Medications can block the enzyme that breaks down a neurotransmitter so that more of it reaches nerve receptors.
Example: Donepezil, galantamine and rivastigmine block the enzyme acetylcholinesterase, which breaks down the neurotransmitter acetylcholine. These medications are used to stabilize and improve memory and cognitive function in people with Alzheimer’s disease, as well as other neurodegenerative disorders.
Medications can block the neurotransmitter from being received at its receptor site.
Example: Selective serotonin reuptake inhibitors are a type of drug class that blocks serotonin from being received and absorbed by a nerve cell. These drugs may be helpful in treating depression, anxiety and other mental health conditions.
Medications can block the release of a neurotransmitter from a nerve cell.
Example: Lithium works as a treatment for mania partially by blocking norepinephrine release and is used in the treatment of bipolar disorder.
Additional Information
Neurotransmitters are chemical messengers in the body. Their function is to transmit signals from nerve cells to target cells. These signals help regulate bodily functions ranging from heart rate to appetite.
Neurotransmitters are part of the nervous system. They play a crucial role in human development and many bodily functions.
What is a neurotransmitter?
The nervous system controls the body’s organs and plays a role in nearly all bodily functions. Nerve cells, also known as neurons, and their neurotransmitters play important roles in this system.
Nerve cells fire nerve impulses. They do this by releasing neurotransmitters, also known as the body’s chemical messengers. These chemicals carry signals to other cells.
Neurotransmitters relay their messages by traveling between cells and attaching to specific receptors on target cells.
Each neurotransmitter attaches to a different receptor. For example, dopamine molecules attach to dopamine receptors. When they attach, it triggers an action in the target cells.
After neurotransmitters deliver their messages, the body breaks them down or recycles them.
What do neurotransmitters do?
The brain needs neurotransmitters to regulate many necessary functions, including:
* heart rate
* breathing
* sleep cycles
* digestion
* mood
* concentration
* appetite
* muscle movement
Neurotransmitters also play a role in early human development.
Types of neurotransmitters
Experts have identified over 100 neurotransmitters to date and are still discovering more.
Neurotransmitters have different types of actions:
* Excitatory neurotransmitters encourage a target cell to take action.
* Inhibitory neurotransmitters decrease the chances of the target cell taking action. In some cases, these neurotransmitters have a relaxation-like effect.
* Modulatory neurotransmitters can send messages to many neurons at the same time. They also communicate with other neurotransmitters.
Some neurotransmitters can carry out several functions depending on the type of receptor they connect to.

#75 Jokes » Miscellaneous Food Jokes - II » 2026-03-06 00:03:32
- Jai Ganesh
- Replies: 0
Q: Why did the skittles go to school?
A: Because they wanted to be smarties!
* * *
Q: How do you make a fruit punch?
A: Give it boxing lessons.
* * *
Q: Did you hear about the angry pancake?
A: He just flipped.
* * *
Q: Why did the pecans run across the busy road?
A: Because they were nuts!
* * *
Q: What is pink, goes in hard and dry and comes out soft and wet?
A: Bubble Gum.
* * *