Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
Pages: 1
#1 Yesterday 17:40:56
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,537
Silver Nitrate
Silver Nitrate
Gist
Silver nitrate is a colorless, odorless inorganic compound commonly used as a versatile chemical precursor in photography, electroplating, and for manufacturing mirrors. It is widely used in medicine as a topical antiseptic and cauterizing agent for wound care and warts. It is prepared by dissolving silver in nitric acid and must be handled with care due to its corrosive nature.
Silver nitrate is used medically as an antiseptic, antibacterial, and cauterizing agent for wound care, wart removal, and controlling bleeding by releasing silver ions that kill microbes and form protective scabs, while industrially it serves as a precursor for silver compounds, in photographic emulsions, and for analytical chemistry (like testing for halides). It is applied topically to treat wounds, burns, and skin tags, though its use in newborns for eye infections is largely replaced by other treatments in the U.S.
Summary
Details
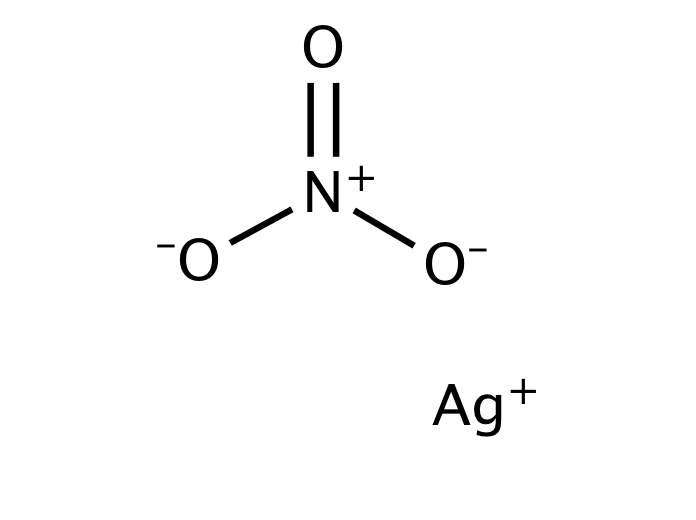
Silver nitrate is an inorganic compound with chemical formula AgNO3. It is a versatile precursor to many other silver compounds, such as those used in photography. It is far less sensitive to light than the halides.[citation needed] It was once called lunar caustic because silver was called luna by ancient alchemists who associated silver with the moon. In solid silver nitrate, the silver ions are three-coordinated in a trigonal planar arrangement.
Uses:
Precursor to other silver compounds
Silver nitrate is the least expensive salt of silver; it offers several other advantages as well. It is non-hygroscopic, in contrast to silver fluoroborate and silver perchlorate. In addition, it is relatively stable to light, and it dissolves in numerous solvents, including water. The nitrate can be easily replaced by other ligands, rendering AgNO3 versatile. Treatment with solutions of halide ions gives a precipitate of AgX (X = Cl, Br, I). When making photographic film, silver nitrate is treated with halide salts of sodium or potassium to form insoluble silver halide in situ in photographic gelatin, which is then applied to strips of tri-acetate or polyester. Similarly, silver nitrate is used to prepare some silver-based explosives, such as the fulminate, azide, or acetylide, through a precipitation reaction.
Halide abstraction
The silver cation, Ag+, reacts quickly with halide sources to produce the insoluble silver halide, which is a cream precipitate if Br− is used, a white precipitate if Cl− is used and a yellow precipitate if I− is used. This reaction is commonly used in inorganic chemistry to abstract halides.
Other silver salts with non-coordinating anions, namely silver tetrafluoroborate and silver hexafluorophosphate are used for more demanding applications.
Similarly, this reaction is used in analytical chemistry to confirm the presence of chloride, bromide, or iodide ions. Samples are typically acidified with dilute nitric acid to remove interfering ions, e.g. carbonate ions and sulfide ions. This step avoids confusion of silver sulfide or silver carbonate precipitates with that of silver halides. The color of precipitate varies with the halide: white (silver chloride), pale yellow/cream (silver bromide), yellow (silver iodide). AgBr and especially AgI photo-decompose to the metal, as evidenced by a grayish color on exposed samples.
The same reaction was used on steamships in order to determine whether or not boiler feedwater had been contaminated with seawater. It is still used to determine if moisture on formerly dry cargo is a result of condensation from humid air, or from seawater leaking through the hull.
Organic synthesis
Silver nitrate is used in many ways in organic synthesis, e.g. for deprotection and oxidations. Ag+ binds alkenes reversibly, and silver nitrate has been used to separate mixtures of alkenes by selective absorption. The resulting adduct can be decomposed with ammonia to release the free alkene. Silver nitrate is highly soluble in water but is poorly soluble in most organic solvents, except acetonitrile (111.8 g/100 g, 25 °C).
Biology
In histology, silver nitrate is used for silver staining, for demonstrating reticular fibers, proteins and nucleic acids. For this reason it is also used to demonstrate proteins in polyacrylamide gel electrophoresis (PAGE) gels. It can be used as a stain in scanning electron microscopy.
Cut flower stems can be placed in a silver nitrate solution, which prevents the production of ethylene. This delays ageing of the flower.
Indelible ink
Silver nitrate produces long-lasting stain when applied to skin and is one of indelible ink’s ingredients. An electoral stain makes use of this to mark a finger of people who have voted in an election, allowing easy identification to prevent double-voting.
In addition to staining skin, silver nitrate has a history of use in stained glass. In the 14th century, artists began using a "silver stain" (also known as a yellow stain) made from silver nitrate to create a yellow effect on clear glass. The stain would produce a stable color that could range from pale lemon to deep orange or gold. Silver stain was often used with glass paint, and was applied to the opposite side of the glass as the paint. It was also used to create a mosaic effect by reducing the number of pieces of glass in a window. Despite the age of the technique, this process of creating stained glass remains almost entirely unchanged.
Medicine:
Silver salts have antiseptic properties. In 1881 Credé introduced a method known as Credé's prophylaxis, which used of dilute (2%) solutions of silver nitrate in newborn babies' eyes at birth to prevent contraction of gonorrhea from the mother, which could cause blindness via ophthalmia neonatorum. (Modern antibiotics are now used instead).
Fused silver nitrate, shaped into sticks, was traditionally called "lunar caustic". It is used as a cauterizing agent, for example to remove granulation tissue around a stoma. General Sir James Abbott noted in his journals that in India in 1827 it was infused by a British surgeon into wounds in his arm resulting from the bite of a mad dog to cauterize the wounds and prevent the onset of rabies.
Silver nitrate is used to cauterize superficial blood vessels in the nose to help prevent nosebleeds.
Dentists sometimes use silver nitrate-infused swabs to heal oral ulcers. Silver nitrate is used by some podiatrists to kill cells located in the nail bed.
The Canadian physician C. A. Douglas Ringrose researched the use of silver nitrate for sterilization procedures, believing that silver nitrate could be used to block and corrode the fallopian tubes. The technique was ineffective.
Disinfection
Much research has been done in evaluating the ability of the silver ion at inactivating Escherichia coli, a microorganism commonly used as an indicator for fecal contamination and as a surrogate for pathogens in drinking water treatment. Concentrations of silver nitrate evaluated in inactivation experiments range from 10–200 micrograms per liter as Ag+. Silver's antimicrobial activity saw many applications prior to the discovery of modern antibiotics, when it fell into near disuse. Its association with argyria made consumers wary and led them to turn away from it when given an alternative.
Against warts
Repeated daily application of silver nitrate can induce adequate destruction of cutaneous warts, but occasionally pigmented scars may develop. In a placebo-controlled study of 70 patients, silver nitrate given over nine days resulted in clearance of all warts in 43% and improvement in warts in 26% one month after treatment compared to 11% and 14%, respectively, in the placebo group.
Safety
As an oxidant, silver nitrate should be properly stored away from organic compounds. It reacts explosively with ethanol. Despite its common usage in extremely low concentrations to prevent gonorrhea and control nosebleeds, silver nitrate is still very toxic and corrosive. Brief exposure will not produce any immediate side effects other than the purple, brown or black stains on the skin, but upon constant exposure to high concentrations, side effects will be noticeable, which include burns. Long-term exposure may cause eye damage. Silver nitrate is known to be a skin and eye irritant. Silver nitrate has not been thoroughly investigated for potential carcinogenic effect.
Silver nitrate is currently unregulated in water sources by the United States Environmental Protection Agency. However, if more than 1 gram of silver is accumulated in the body, a condition called argyria may develop. Argyria is a permanent cosmetic condition in which the skin and internal organs turn a blue-gray color. The United States Environmental Protection Agency used to have a maximum contaminant limit for silver in water until 1990, when it was determined that argyria did not impact the function of any affected organs despite the discolouration. Argyria is more often associated with the consumption of colloidal silver solutions rather than with silver nitrate, since it is only used at extremely low concentrations to disinfect the water. However, it is still important to be wary before ingesting any sort of silver-ion solution.
Additional Information
Silver nitrate is a chemical compound with the formula AgNO3. It consists of an ionic bond between the silver cation (Ag+) and the nitrate anion (NO3–). Due to the ionic nature of this compound, it readily dissolves in water and dissociates into its constituent ions.
Silver nitrate is a precursor to many compounds of silver, including the silver compounds used in photography. When compared to silver halides, which are used in photography due to their sensitivity to light, AgNO3 is quite stable when exposed to light.
Structure of AgNO3
The nitrate ion described above consists of one nitrogen atom which is surrounded by three oxygen atoms. The nitrogen-oxygen bonds in this ion are similar to each other. The formal charge assigned to the nitrogen atom is -1, whereas each oxygen atom holds a charge of -⅔. The net charge associated with the nitrate ion is -1, which is quenched by the +1 charge held by the Ag+ ion via an ionic bond in AgNO3. It can be noted that the structure of the nitrate ion is stabilized by resonance.
Properties of Silver Nitrate
Some important physical and chemical properties of silver nitrate are listed in this subsection.
Physical Properties
* The molar mass of silver nitrate is 169.872 grams per mole.
* AgNO3 has a colourless appearance in its solid-state and is odourless.
* In its solid state, it has a density of 4.35 grams per cubic centimetre. Its density in the liquid state at a temperature of 210 degrees C corresponds to 3.97 g/{cm}^{3}.
* The melting and boiling points of silver nitrate are 482.8 K and 713 K respectively. However, this compound tends to decompose at temperatures approaching its boiling point.
* Silver nitrate, like most ionic compounds, dissolves readily in water. Its solubility in water corresponds to 122 g /100mL at 0 oC and 256g / 100mL at a temperature of 25 degrees centigrade.
* The crystal structure of AgNO3 is orthorhombic.
Chemical Properties
* The hazards of AgNO3 include its toxic and corrosive nature.
* The reaction between silver nitrate and ethanol is explosive.
* The silver present in the silver nitrate compound is displaced by copper, which forms copper nitrate. The chemical equation for this reaction is given by
2AgNO3 + Cu → Cu(NO3)2 + 2Ag
* When heated to 440 oC, this compound completely decomposes to give oxygen, nitrogen dioxide, and silver.
* Silver nitrate on decomposition gives silver, oxygen gas and nitrite.
* It can be noted that even though metal nitrates generally decompose to yield metal oxides, the decomposition reaction of silver nitrate gives rise to elemental silver because silver oxide decomposes at an even lower temperature than AgNO3.
Uses of Silver Nitrate
Silver nitrate has a wide range of applications in many fields such as biology, chemical synthesis, and medicine. Some of these uses of AgNO3 are listed below.
* Silver nitrate is a very versatile compound because the nitrate ion can be replaced by other ligands that can bind to the silver ion.
* Due to the ability of this compound to form a precipitate of silver halides when treated with halide ions, it is used while making photographic films.
* Many silver-based explosives can be prepared with a precipitation reaction of silver nitrate.
* In the field of inorganic chemistry, halides are extracted with the help of this compound.
* The branch of chemistry known as analytical chemistry uses this reaction to check for the presence of halide anions such as the iodide, bromide, or chloride ions.
* Mixtures of alkenes can be separated with the help of this compound since the silver cation binds with alkenes in a reversible fashion.
* When diluted with water to a concentration of 0.5%, silver nitrate can serve as an antiseptic in many medical setups.
* A diluted solution of AgNO3 can be administered to the eyes of a baby which is born to a mother suffering from gonorrhea, which combats the gonorrhoea bacteria and protects the baby from the onset of blindness.
* This compound is also known to be used for the treatment and the removal of unwanted warts in human beings.
Frequently Asked Questions:
What are the uses of silver nitrate?
Silver nitrate is widely used in many organic synthesis reactions in several ways. For example, for the deprotection and oxidation reactions. The Ag+ ion reversibly binds alkenes, and selectively adsorbing silver nitrate can be used to isolate alkene mixtures. The resulting adduct can be decomposed (in order to release the free alkene) with ammonia. Silver nitrate has been, in the past, used for silver staining (a process that employs silver or silver compounds to selectively change the appearance of a specific object). This compound is also used in medicine owing to its antiseptic qualities.
Is silver nitrate dangerous?
Silver nitrate is an oxidant and must, therefore, be kept away from organic compounds. Despite its widespread usage (especially in extremely low amounts) for the prevention of gonorrhoea and to stop bleeding from the nose, silver nitrate is often highly toxic and corrosive. Short-term exposure to this compound does not cause any immediate side effects apart from the development of a violet, brown or black stain on the part of the skin that was in contact with the silver nitrate. However, exposure to this compound over long periods of time is often accompanied by damage to the eyes. This compound is widely classified as an irritant to the skin and the eyes.
How is silver nitrate prepared?
Silver nitrate is usually prepared by combining silver with nitric acid. Common silver objects used in these reactions include silver bullions and silver foils. The products formed in this reaction include silver nitrate, water, and nitrogen oxides. The by-products of this chemical reaction depend on the nitric acid concentration that is used. It is important to note that this reaction must be carried out under a fume hood because of the evolution of poisonous oxides of nitrogen during the chemical reaction.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1