Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
Pages: 1
#1 Yesterday 17:11:47
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,563
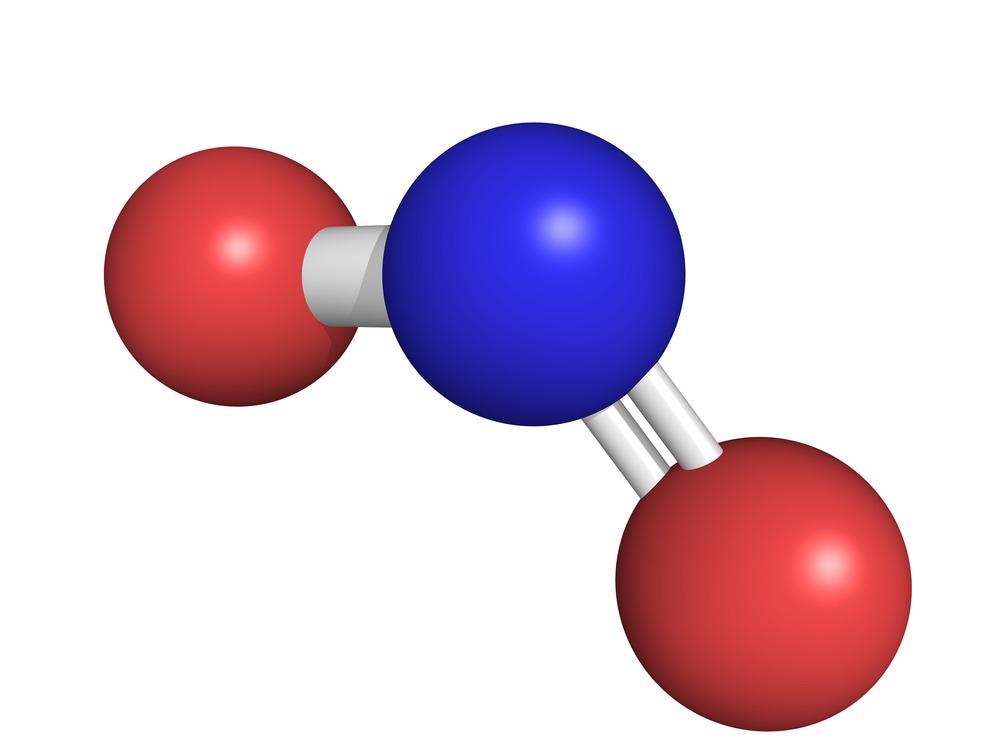
Nitrogen Dioxide
Nitrogen Dioxide
Gist
Nitrogen dioxide is a highly reactive, reddish-brown toxic gas with a pungent odor, acting as a major air pollutant and strong oxidant. Primarily produced by burning fossil fuels in vehicles, power plants, and industrial equipment, it poses severe respiratory health risks, including asthma, and contributes to smog, acid rain, and ozone formation.
NO2 is comprised of one atom of nitrogen and two atoms of oxygen, and is a gas at ambient temperatures. It has a pungent smell, and is brownish red in color.
Summary
Nitrogen dioxide is a chemical compound with the formula NO2. One of several nitrogen oxides, nitrogen dioxide is a reddish-brown gas. It is a paramagnetic, bent molecule with C2v point group symmetry. Industrially, NO2 is an intermediate in the synthesis of nitric acid, millions of tons of which are produced each year, primarily for the production of fertilizers.
Nitrogen dioxide is poisonous and can be fatal if inhaled in large quantities. Cooking with a gas stove produces nitrogen dioxide which causes poorer indoor air quality. Combustion of gas can lead to increased concentrations of nitrogen dioxide throughout the home environment which is linked to respiratory issues and diseases. The LC50 (median lethal dose) for humans has been estimated to be 174 ppm for a 1-hour exposure. It is also included in the NOx family of atmospheric pollutants.
Properties
Nitrogen dioxide is a reddish-brown gas with a pungent, acrid odor above 21.2 °C (70.2 °F; 294.3 K) and becomes a yellowish-brown liquid below 21.2 °C (70.2 °F; 294.3 K). It forms an equilibrium with its dimer, dinitrogen tetroxide (N2O4), and converts almost entirely to N2O4 below −11.2 °C (11.8 °F; 261.9 K).
The bond length between the nitrogen atom and the oxygen atom is 119.7 pm. This bond length is consistent with a bond order between one and two. Nitrogen dioxide is a doublet state.
Details
Nitrogen dioxide, or NO2, is a gaseous air pollutant composed of nitrogen and oxygen and is one of a group of related gases called nitrogen oxides, or NOx. Nitrogen dioxide forms when fossil fuels such as coal, oil, methane gas (natural gas) or diesel are burned at high temperatures. NO2 and other nitrogen oxides in the outdoor air contribute to particle pollution and to the chemical reactions that make ozone. It is one of six widespread air pollutants for which there are national air quality standards to limit their levels in the outdoor air. NO2 can also form indoors when fuels like wood or gas are burned.
What Are the Health Effects of Nitrogen Dioxide Pollution?
Nitrogen dioxide causes a range of harmful effects on the lungs, including:
* Increased inflammation of the airways;
* Worsened cough and wheezing;
* Reduced lung function;
* Increased asthma attacks; and
* Greater likelihood of emergency department and hospital admissions.
Scientific evidence suggests that exposure to NO2 could likely cause asthma in children.
A 2022 review of multiple studies found that elevated levels of NO2, as well as elevated particulate matter and sulfur dioxide, were strongly associated with heart and lung harm, affected pregnancy and birth outcomes, and were likely associated with increased risk of kidney and neurological harm, autoimmune disorders and cancer.
What Are the Sources of Nitrogen Dioxide Emissions?
As of 2020, human-made sources in the U.S. emit 7.64 million short tons of nitrogen oxides per year (down from 15 million short tons per year in 2011) mainly from burning fuels. Trucks, buses, and cars are the largest sources of NO2 emissions, followed by diesel-powered non-road equipment, industrial processes such as oil and gas production, industrial boilers and other movable engines, and coal-fired power plants. Emissions of nitrogen dioxide will decline as cleanup of many of these sources continue in future years.
Where Do High NO2 Concentrations Occur?
Monitors show the highest concentrations of outdoor NO2 in large urban regions such as the Northeast corridor, Chicago and Los Angeles. Levels are higher on or near heavily traveled roadways.
It is important to note that NO2 and other nitrogen oxides are also produced from burning natural gas (methane), both outdoors and indoors. Outdoors, this can include gas-fired power plants and from facilities that extract, process or transport oil and gas if they burn it in flares or to power equipment. Indoors, appliances such as stoves, dryers and space heaters that burn natural gas, liquified petroleum gas (or LPG, which includes propane and butane) and kerosene can produce substantial amounts of nitrogen dioxide. If those appliances are not fully vented to the outside, levels of NO2 can build up to unhealthy levels indoors.
Who Is at Risk?
While everyone is at risk from health impacts of nitrogen dioxide pollution, those that live near the emission sources are at higher risk. Other vulnerable subpopulations at higher risk from nitrogen dioxide exposure include:
* Individuals who are pregnant;
* Infants, children and teens;
* Older adults (>65 years of age);
* People with pre-existing medical conditions such as asthma, chronic obstructive pulmonary disease (COPD), cardiovascular disease, diabetes, lung cancer
* Current or former smokers;
* People with low socioeconomic status; and
* People of color.
What Can We Do about It?
The good news is that for much of the nation, the outdoor air has much lower levels of nitrogen dioxide now than in previous decades. Under the federal Clean Air Act, more protective standards nationwide have helped drive down nitrogen dioxide emissions. Power plants, industrial sites and on-road vehicles are cleaner than they used to be, which has driven nationwide improvement in air quality. However, far too many people still breathe in unhealthy levels of nitrogen dioxide pollution.
Individuals can take steps to protect themselves on days with unhealthy levels of air pollutants and also ask policymakers at all levels of government to continue to require cleanup of air pollution.
Additional Information:
What is Nitrogen dioxide?
NO2 is a highly poisonous gas with the chemical name Nitrogen dioxide.
It is also called Nitrogen (IV) oxide or Deutoxide of nitrogen. It is one of the major atmospheric pollutants that absorb UV light and stops to reach the earth’s surface.
Nitrogen (IV) oxide is a yellowish-brown liquid in its compressed form or reddish-brown gas. Its vapours are heavier when compared to air.
Nitrogen dioxide Sources – NO2
Over 98 percent of man-made N0, emissions result from Combustion with the majority due to stationary sources. Combustion generated oxides of nitrogen are emitted predominantly as nitric oxide, N0, a relatively harmless gas, but one which is rapidly converted in the atmosphere to the toxic nitrogen dioxide.
Nitrogen dioxide is also a precursor in the formation of nitrate serosols and nitrosamines, the health effects of which are under study. Because of the quantity generated and their potential for widespread adverse effects on public health and welfare, nitrogen oxides are among the atmospheric pollutants for which standards and regularly controls have been established both by the U.S. c
NO2 Uses (Nitrogen dioxide)
* Nitrogen dioxide is used as an intermediate in the production of nitric acid.
* Used in the manufacturing of oxidized cellulose compounds.
* Used as a catalyst.
* Used as an intermediate in the production of sulphuric acid.
* Used as an oxidizer for rocket fuels.
* Used as a nitrating agent.
* Used to bleach flour.
* Used as an oxidizing agent.
Health Hazards
Severe exposures of Deutoxide of nitrogen can be fatal. When in contact it causes a burning sensation in the eyes and skin. When in liquid form it causes frostbite. It is reported to react with the blood to form methemoglobin. When heated to decompose, it releases toxic fumes of nitrogen oxides.
Nitrogen dioxide is an irritant gas which causes inflammation of the airways at high concentrations. NO2 mainly affects respiratory conditions which cause high levels of airway inflammation. Long-term exposure will decrease lung capacity, increase the probability of respiratory problems and increase allergy response. NO2 also contributes to the production of small particles (PM) and ozone at ground level, both associated with harmful effects on the environment.
Frequently Asked Questions – FAQs
Q1: What is nitrogen dioxide used for?
A1: Nitrogen Dioxide, NO2; was used as a catalyst in some oxidation reactions; as an inhibitor to prevent the polymerization of acrylates during distillation; as an organic compound nitrating agent; as an oxidizing agent; as a rocket fuel; as a flour bleaching agent.
Q2: What does nitrogen dioxide do?
A2: The key consequence of breathing in elevated nitrogen dioxide levels is an increased risk of respiratory disorders. Nitrogen dioxide inflames the lungs ‘ lining and can decrease immunity to infections of the lungs. This can lead to wheezing, coughing, colds, pneumonia, and bronchitis issues.
Q3: Does nitrogen dioxide cause global warming?
A3: Nitric oxide and nitrogen dioxide are the two most toxic and dangerous nitrogen oxides. Nitrous oxide, often referred to as laughing gas, is a greenhouse gas which contributes to global warming.
Q4: What are the main sources of nitrogen oxides?
A4: Natural causes include volcanoes, rivers, biological collapse and bolts of lightning. Every year human activities add 24 million tons of nitrogen oxides to our atmosphere.
Q5: Is nitrogen dioxide heavier than air?
A5: At high concentrations, nitric oxide is quickly oxidized into the air to produce nitrogen dioxide. Expositions. Nitrogen dioxide is heavier than air, so exposure can result in asphyxiation in poorly ventilated, sealed, or low-lying areas. The gasses at room temperature are both nitrogen dioxide and nitric oxide.
Q6: What is nitrogen dioxide made from?
A6: Nitrogen dioxide is an irritant gas which causes inflammation of the airways at high concentrations. As nitrogen is released during the combustion of fuel it reacts with atoms of oxygen to produce nitric oxide (NO). It additionally reacts with oxygen to form nitrogen dioxide (NO2).
Q7: What causes high nitrogen dioxide levels?
A7: Nitrogen dioxide comes from cars, power stations, and commercial and off-road outlets such as building machinery, lawn machines, and gardening. Such outlets both use fossil fuels. People living or working close to busy highways can suffer high exposure.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1