Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
- Index
- » Science HQ
- » Cobalt
Pages: 1
#1 2025-07-27 15:51:44
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,769
Cobalt
Cobalt
Gist
Cobalt is a versatile metal used in various applications, most notably in rechargeable batteries, high-strength alloys, and pigments. It is also crucial in medical treatments, industrial catalysts, and even as a component of vitamin B12.
Classified by the EU and the US as a Critical Raw Material, cobalt is recognized as an important technology-enabling metal where energy storage, high temperature resilience, hardness, process efficiency and environmental benefits are required.
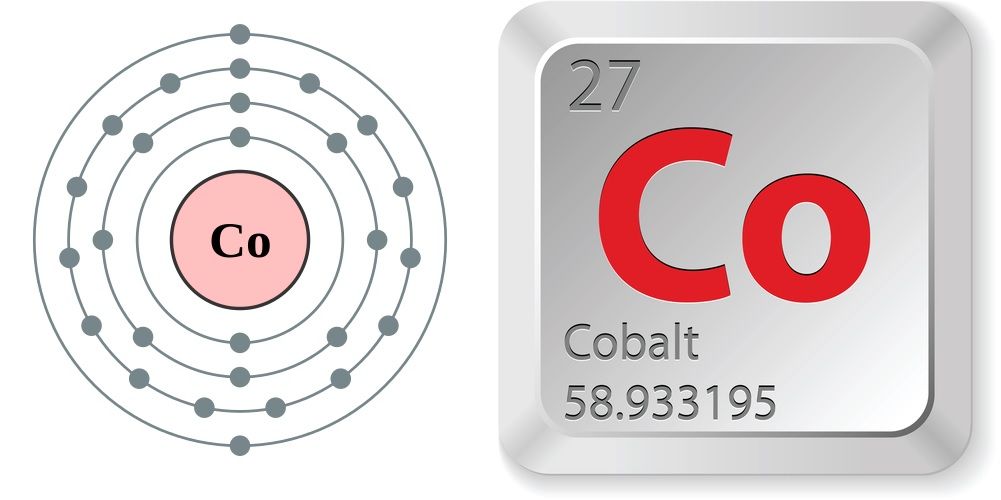
Cobalt is a chemical element with the symbol Co and atomic number 27. It is a hard, lustrous, silver-grey transition metal, known for its magnetic properties (ferromagnetic) and ability to withstand high temperatures. Cobalt is a d-block element and is found in the Earth's crust primarily in combined forms, with small deposits in meteoric iron.
Summary
Cobalt is a chemical element; it has symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, produced by reductive smelting, is a hard, lustrous, somewhat brittle, gray metal.
Cobalt-based blue pigments (cobalt blue) have been used since antiquity for jewelry and paints, and to impart a distinctive blue tint to glass. The color was long thought to be due to the metal bismuth. Miners had long used the name kobold ore (German for goblin ore) for some of the blue pigment-producing minerals. They were so named because they were poor in known metals and gave off poisonous math-containing fumes when smelted. In 1735, such ores were found to be reducible to a new metal (the first discovered since ancient times), which was ultimately named for the kobold.
Today, cobalt is usually produced as a by-product of copper and nickel mining, but sometimes also from one of a number of metallic-lustered ores such as cobaltite (CoAsS). The Copperbelt in the Democratic Republic of the Congo (DRC) and Zambia yields most of the global cobalt production. World production in 2016 was 116,000 tonnes (114,000 long tons; 128,000 short tons) according to Natural Resources Canada, and the DRC alone accounted for more than 50%. In 2024, production exceeded 300,000 tons, of which DRC accounted for more than 80%.
Cobalt is primarily used in lithium-ion batteries, and in the manufacture of magnetic, wear-resistant and high-strength alloys. The compounds cobalt silicate and cobalt(II) aluminate (CoAl2O4, cobalt blue) give a distinctive deep blue color to glass, ceramics, inks, paints and varnishes. Cobalt occurs naturally as only one stable isotope, cobalt-59. Cobalt-60 is a commercially important radioisotope, used as a radioactive tracer and for the production of high-energy gamma rays. Cobalt is also used in the petroleum industry as a catalyst when refining crude oil. This is to purge it of sulfur, which is very polluting when burned and causes acid rain.
Cobalt is the active center of a group of coenzymes called cobalamins. Vitamin B12, the best-known example of the type, is an essential vitamin for all animals. Cobalt in inorganic form is also a micronutrient for bacteria, algae, and fungi.
The name cobalt derives from a type of ore considered a nuisance by 16th century German silver miners, which in turn may have been named from a spirit or goblin held superstitiously responsible for it; this spirit is considered equitable to the kobold (a household spirit) by some, or, categorized as a gnome (mine spirit) by others.
Details
Cobalt (Co), chemical element, ferromagnetic metal of Group 9 (VIIIb) of the periodic table, used especially for heat-resistant and magnetic alloys.
The metal was isolated (c. 1735) by Swedish chemist Georg Brandt, though cobalt compounds had been used for centuries to impart a blue colour to glazes and ceramics. Cobalt has been detected in Egyptian statuettes and Persian necklace beads of the 3rd millennium bce, in glass found in the Pompeii ruins, and in China as early as the Tang dynasty (618–907 ce) and later in the blue porcelain of the Ming dynasty (1368–1644). The name kobold was first applied (16th century) to ores thought to contain copper but eventually found to be poisonous math-bearing cobalt ores. Brandt finally determined (1742) that the blue colour of those ores was due to the presence of cobalt.
Element Properties
atomic number : 27
atomic weight : 58.933194
melting point : 1,495 °C (2,723 °F)
boiling point : 2,870 °C (5,198 °F)
density : 8.9 gram/{cm}^{3} at 20 °C (68 °F)
oxidation states : +2, +3.
Occurrence, properties, and uses
Cobalt, though widely dispersed, makes up only 0.001 percent of Earth’s crust. It is found in small quantities in terrestrial and meteoritic native nickel-iron, in the Sun and stellar atmospheres, and in combination with other elements in natural waters, in ferromanganese crusts deep in the oceans, in soils, in plants and animals, and in minerals such as cobaltite, linnaeite, skutterudite, smaltite, heterogenite, and erythrite. In animals, cobalt is a trace element essential in the nutrition of ruminants (cattle, sheep) and in the maturation of human red blood cells in the form of vitamin B12, the only vitamin known to contain such a heavy element.
With few exceptions, cobalt ore is not usually mined for the cobalt content. Rather, it is often recovered as a by-product from the mining of ores of iron, nickel, copper, silver, manganese, zinc, and math, which contain traces of cobalt. Complex processing is required to concentrate and extract cobalt from these ores. By the second decade of the 21st century, the Democratic Republic of the Congo (DRC), China, Canada, and Russia were the world’s leading producers of mined cobalt. The largest producer of refined cobalt, however, was China, which imported vast additional amounts of cobalt mineral resources from the DRC. (For additional information on the mining, refining, and recovery of cobalt, see cobalt processing.)
Polished cobalt is silver-white with a faint bluish tinge. Two allotropes are known: the hexagonal close-packed structure, stable below 417 °C (783 °F), and the face-centred cubic, stable at high temperatures. It is ferromagnetic up to 1,121 °C (2,050 °F, the highest known Curie point of any metal or alloy) and may find application where magnetic properties are needed at elevated temperatures.
Cobalt is one of the three metals that are ferromagnetic at room temperature. It dissolves slowly in dilute mineral acids, does not combine directly with either hydrogen or nitrogen, but will combine, on heating, with carbon, phosphorus, or sulfur. Cobalt is also attacked by oxygen and by water vapour at elevated temperatures, with the result that cobaltous oxide, CoO (with the metal in the +2 state), is produced.
Natural cobalt is all stable isotope cobalt-59, from which the longest-lived artificial radioactive isotope cobalt-60 (5.3-year half-life) is produced by neutron irradiation in a nuclear reactor. Gamma radiation from cobalt-60 has been used in place of X-rays or alpha rays from radium in the inspection of industrial materials to reveal internal structure, flaws, or foreign objects. It has also been used in cancer therapy, in sterilization studies, and in biology and industry as a radioactive tracer.
Most of the cobalt produced is used for special alloys. A relatively large percentage of the world’s production goes into magnetic alloys such as the Alnicos for permanent magnets. Sizable quantities are utilized for alloys that retain their properties at high temperatures and superalloys that are used near their melting points (where steels would become too soft). Cobalt is also employed for hard-facing alloys, tool steels, low-expansion alloys (for glass-to-metal seals), and constant-modulus (elastic) alloys (for precision hairsprings). Cobalt is the most satisfactory matrix for cemented carbides.
Finely divided cobalt ignites spontaneously. Larger pieces are relatively inert in air, but above 300 °C (570 °F) extensive oxidation occurs.
Compounds
In its compounds cobalt nearly always exhibits a +2 or +3 oxidation state, although states of +4, +1, 0, and −1 are known. The compounds in which cobalt exhibits the +2 oxidation state (Co2+, the ion being stable in water) are called cobaltous, while those in which cobalt exhibits the +3 oxidation state (Co3+) are called cobaltic.
Both Co2+ and Co3+ form numerous coordination compounds, or complexes. Co3+ forms more known complex ions than any other metal except platinum. The coordination number of the complexes is generally six.
Cobalt forms two well-defined binary compounds with oxygen: cobaltous oxide, CoO, and tricobalt textroxide, or cobalto-cobaltic oxide, Co3O4. The latter contains cobalt in both +2 and +3 oxidation states and constitutes up to 40 percent of the commercial cobalt oxide used in the manufacture of ceramics, glass, and enamel and in the preparation of catalysts and cobalt metal powder.
One of the more important salts of cobalt is the sulfate CoSO4, which is employed in electroplating, in preparing drying agents, and for pasture top-dressing in agriculture. Other cobaltous salts have significant applications in the production of catalysts, driers, cobalt metal powders, and other salts. Cobaltous chloride (CoCl2∙6H2O in commercial form), a pink solid that changes to blue as it dehydrates, is utilized in catalyst preparation and as an indicator of humidity. Cobaltous phosphate, Co3(PO4)2∙8H2O, is used in painting porcelain and colouring glass.
Additional Information:
Appearance
A lustrous, silvery-blue metal. It is magnetic.
Uses
Cobalt, like iron, can be magnetised and so is used to make magnets. It is alloyed with aluminium and nickel to make particularly powerful magnets.
Other alloys of cobalt are used in jet turbines and gas turbine generators, where high-temperature strength is important.
Cobalt metal is sometimes used in electroplating because of its attractive appearance, hardness and resistance to corrosion.
Cobalt salts have been used for centuries to produce brilliant blue colours in paint, porcelain, glass, pottery and enamels.
Radioactive cobalt-60 is used to treat cancer and, in some countries, to irradiate food to preserve it.
Biological role
Cobalt is an essential trace element, and forms part of the active site of vitamin B12. The amount we need is very small, and the body contains only about 1 milligram. Cobalt salts can be given to certain animals in small doses to correct mineral deficiencies. In large doses cobalt is carcinogenic.
Cobalt-60 is a radioactive isotope. It is an important source of gamma-rays. It is widely used in cancer treatment, as a tracer and for radiotherapy.
Natural abundance
Cobalt is found in the minerals cobaltite, skutterudite and erythrite. Important ore deposits are found in DR Congo, Canada, Australia, Zambia and Brazil. Most cobalt is formed as a by-product of nickel refining.
A huge reserve of several transition metals (including cobalt) can be found in strange nodules on the floors of the deepest oceans. The nodules are manganese minerals that take millions of years to form, and together they contain many tonnes of cobalt.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1
- Index
- » Science HQ
- » Cobalt