Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
#2726 2026-03-18 00:04:18
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,617
Re: Miscellany
2525) Sulfur Dioxide
Gist
Sulfur dioxide (SO2) is a colorless, pungent, and toxic gas composed of sulfur and oxygen, primarily produced by burning fossil fuels and volcanic activity. It is a major air pollutant known to cause respiratory issues, such as difficulty breathing and asthma exacerbation. Industrially, it is crucial for manufacturing sulfuric acid and used as a preservative in food and wine.
Sulfur dioxide (SO2) is an industrial chemical used primarily as a precursor for sulfuric acid production, a preservative (especially for dried fruits and wine), a bleaching agent in paper/pulp manufacturing, and a disinfectant. It acts as a reducing agent in chemical processes and a refrigerant in industrial cooling systems.
Summary
Sulfur dioxide (SO2), is an inorganic compound, a heavy, colorless, poisonous gas. It is produced in huge quantities in intermediate steps of sulfuric acid manufacture.
Sulfur dioxide has a pungent, irritating odor, familiar as the smell of a just-struck match. Occurring in nature in volcanic gases and in solution in the waters of some warm springs, sulfur dioxide usually is prepared industrially by the burning in air or oxygen of sulfur or such compounds of sulfur as iron pyrite or copper pyrite. Large quantities of sulfur dioxide are formed in the combustion of sulfur-containing fuels.
Sulfur dioxide pollution carries serious health and environmental risks and is one of the six criteria air pollutants regulated by the U.S. Environmental Protection Agency and other regulatory agencies around the world. In the atmosphere sulfur dioxide can combine with water vapor to form sulfuric acid, a major component of acid rain; in the second half of the 20th century, measures to control acid rain were widely adopted. Most of the sulfur dioxide released into the environment comes from coal-fired power plants and petroleum refineries. Paper pulp manufacturing, cement manufacturing, and metal smelting and processing facilities are other important sources.
Sulfur dioxide is a precursor of the trioxide (SO3) used to make sulfuric acid. In the laboratory the gas may be prepared by reducing sulfuric acid (H2SO4) to sulfurous acid (H2SO3), which decomposes into water and sulfur dioxide, or by treating sulfites (salts of sulfurous acid) with strong acids, such as hydrochloric acid, again forming sulfurous acid.
Details
Sulfur dioxide (SO2) is a pungent, toxic gas that is the primary product of burning elemental sulfur. It exists widely in nature, mostly from volcanic activity and burning fossil fuels. It is found elsewhere in the solar system, as a gas in the atmospheres of Venus and Jupiter’s moon Io and as an ice on the other Galilean moons.
The major use of SO2 is in the manufacture of sulfuric acid (H2SO4), the most-produced chemical worldwide. Elemental sulfur and oxygen react to form SO2, which is catalytically oxidized with additional oxygen to make sulfur trioxide (SO3). The SO3 is mixed with existing H2SO4 to produce oleum (fuming sulfuric acid), which is added to water in a strongly exothermic process to make concentrated H2SO4. This is known as the contact process; it dates to an 1831 patent by British inventor Peregrine Phillips.
In chemical laboratories, it has multiple functions, including as a reducing agent, as a reagent in sulfonylation reactions, and as a low-temperature solvent. SO2 is also used to preserve dried fruits such as raisins and prunes and to prevent spoilage in wine.
The hazard information table shows that SO2 is pretty nasty stuff; but, in addition to its value as a chemical, it has another positive side: Volcanoes that emit the gas can have a beneficial effect on climate change. When SO2 spews into the stratosphere, it reacts photochemically with oxygen to form H2SO4 aerosols, which in turn reflect solar radiation and cool the atmosphere. But, as might be expected, even this has a downside because SO2 and H2SO4 contribute to acid rain.
Additional Information:
What Is Sulfur Dioxide?
Sulfur dioxide (SO2) is a gaseous air pollutant composed of sulfur and oxygen. SO2 forms when sulfur-containing fuel such as coal, petroleum oil, or diesel is burned. Sulfur dioxide gas can also change chemically into sulfate particles in the atmosphere, a major part of fine particle pollution, which can blow hundreds of miles away.
What Are the Health Effects of Sulfur Dioxide Pollution?
Sulfur dioxide causes a range of harmful effects on the lungs:
* Wheezing, shortness of breath and chest tightness and other problems, especially during exercise or physical activity. Rapid breathing during exercise helps SO2 reach the lower respiratory tract, as does breathing through the mouth.
* Long-term exposure at high levels increases respiratory symptoms and reduces the ability of the lungs to function.
* Short exposures to peak levels of SO2 in the air can make it difficult for people with asthma to breathe when they are active outdoors.
* Increased risk of hospital admissions or emergency room visits, especially among children, older adults and people with asthma.
What Are the Sources of Sulfur Dioxide Emissions?
As of 2020, human-made sources in the U.S. emit about 1.8 million short tons of sulfur dioxide per year (down from just over 6 million short tons per year in 2011) mainly from burning fuels. Power plants, commercial and institutional boilers, internal combustion engines, manufacturing, and industrial processes such as petroleum refining and metal processing are the largest sources of emissions, followed by diesel engines in old buses and trucks, locomotives, ships, and off-road equipment such as construction vehicles. Emissions of sulfur dioxide will decline as cleanup of many of these sources continue in future years.
Where Do High SO2 Concentrations Occur?
Coal-fired power plants remain one of the biggest sources of sulfur dioxide in the U.S. Columns of emissions (plumes) such as from chimneys of a coal-fired power plant are moved by wind over long distances before touching down at ground level at far away sites. These plumes could also get trapped at the ground level by unusual weather conditions such as a layer of warmer air occurring higher up in the atmosphere (inversion).
Ports, smelters, and other sources of sulfur dioxide also cause high concentrations of emissions nearby.
People who live and work near these large sources get the highest exposure to SO2.
What Can We Do about it?
SO2 levels have improved over time, thanks to policies requiring cleaner fuels and pollution controls on power plants. The nation achieved major reductions in this pollutant through its successful program to reduce acid rain.
However, it remains a health concern. What’s more, even with pollution controls installed, high levels can occur when a polluting source such as a power plant is starting up or shutting down its operation or if its equipment malfunctions.
Individuals can take steps to protect themselves on days with unhealthy levels of air pollutants and also ask policymakers at all levels of government to continue to require cleanup of air pollution.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#2727 2026-03-19 00:08:34
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,617
Re: Miscellany
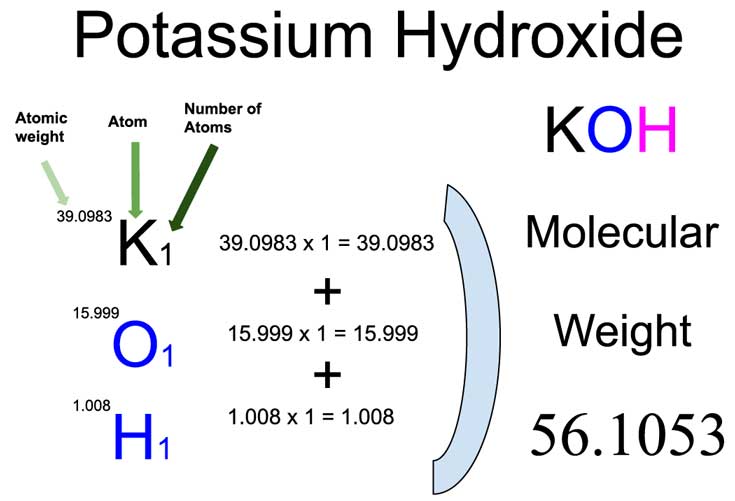
2526) Potassium Hydroxide
Gist
Potassium hydroxide is a disinfectant used to clean hands, skin, and surfaces. Potassium hydroxide, also known as lye is an inorganic compound with the chemical formula KOH. Also commonly referred to as caustic potash, it is a potent base that is marketed in several forms including pellets, flakes, and powders.
Potassium hydroxide is an inorganic compound with the formula KOH, and is commonly called caustic potash. Along with sodium hydroxide, KOH is a prototypical strong base. It has many industrial and niche applications, most of which utilize its caustic nature and its reactivity toward acids.
Summary
Potassium hydroxide is an inorganic compound with the formula KOH, and is commonly called caustic potash.
Along with sodium hydroxide (NaOH), KOH is a prototypical strong base. It has many industrial and niche applications, most of which utilize its caustic nature and its reactivity toward acids. About 2.5 million tonnes were produced in 2023. KOH is noteworthy as the precursor to most soft and liquid soaps, as well as numerous potassium-containing chemicals. It is a white solid that is dangerously corrosive.
Properties and structure
KOH exhibits high thermal stability. Because of this high stability and relatively low melting point, it is often melt-cast as pellets or rods, forms that have low surface area and convenient handling properties. These pellets become tacky in air because KOH is hygroscopic. Most commercial samples are ca. 90% pure, the remainder being water and carbonates. Its dissolution in water is strongly exothermic. Concentrated aqueous solutions are sometimes called potassium lyes. Even at high temperatures, solid KOH does not dehydrate readily.
Details
Potassium hydroxide is also known as caustic potash, lye, and potash lye. This alkali metal hydroxide is a very powerful base. The aqueous form of potassium hydroxide appears as a clear solution. In its solid form, KOH can exist as white to slightly yellow lumps, flakes, pellets, or rods. No characteristic odour can be attributed to this compound in its solid state.
Potassium hydroxide is soluble in water, freely soluble in ethanol, methanol, and glycerin. It is slightly soluble in ether. It is non-combustible but highly corrosive. It is widely used in chemical manufacturing, cleaning compounds, and petroleum refining.
Uses of Potassium Hydroxide
* Potassium hydroxide solution is more conductive when compared to NaOH and therefore used as an electrolyte in some alkaline batteries.
* It is used as a pH control agent in the food industry.
* It is used in the thickening of food.
* It is used in chip fabrication for semiconductors.
* It is used in the manufacturing of cuticle removers which are used in manicure treatment.
* It is used in the identification of species of fungi.
* It is used in mercerizing cotton.
* It is used in alkalimetric titrations in analytical chemistry.
* Used in the manufacturing of liquid fertilisers.
Chemical Reactions Undergone by KOH
1. Saponification of ester
The ester is saponified by heating with a known amount of potassium hydroxide in an organic solvent in a sealed tube. To be useful analytically, this reaction must be quantitative in a reasonable length of time. One condition that favours a rapid and quantitative reaction is the use of KOH as a strong base as possible.
2. KOH reacts with CO2 to produce bicarbonate
The addition of hydroxide ions by adding lime, sodium hydroxide, or potassium hydroxide, adjusts the pH because the hydroxide ion reacts with carbon dioxide to form bicarbonate alkalinity.
Health Hazards of KOH
The health hazards of potassium hydroxide are similar to those of the other strong alkalies, such as sodium hydroxide. Potash lye and its solution can severely irritate skin, mucous membranes, and eyes. When it comes in contact with water or moisture it can generate heat to instigate combustion. Potassium hydroxide is corrosive to tissues.
Frequently Asked Questions – FAQs
Q1: What is potassium hydroxide used for?
A1: Potassium hydroxide, or caustic potash, is used in a wide variety of industries. It is used in the chemical industry, mining, manufacturing of different compounds, fertilisers, in potassium soaps and in detergents.
Q2: What are the dangers of potassium hydroxide?
A2: Causes eye pain, tearing, redness and swelling. Larger exposures cause serious burns with potential subsequent blindness. Chronic exposure: repeated contact with dilute solutions of potassium hydroxide dust has a tissue-destroying effect.
Q3: Is potassium hydroxide a carcinogen?
A3: The National Toxicology Program (NTP), the International Agency for Research on Cancer (IARC), and the Occupational Safety and Health Administration (OSHA) do not recognize potassium hydroxide as a carcinogen. Potassium hydroxide is of low toxicity to marine species.
Q4: What is potassium hydroxide in chemistry?
A4: Potassium hydroxide, also called lye, is an inorganic compound containing the chemical formula KOH. Often commonly called caustic potash, it is a strong base that is sold in different forms including pellets, flakes, and powders.
Q5: What is the pH of KOH?
A5: KOH is an example of a strong base which means that it dissociates completely in an aqueous solution into its ions. Although the pH of KOH or potassium hydroxide is extremely high (typical solutions typically range from 10 to 13), the exact value depends on the concentration of this strong base in water.
Additional Information
Potassium hydroxide (KOH) is an alkali that penetrates and destroys the skin by dissolving keratin. It is used in aqueous solution at concentrations of 5% to 20%, and applied to MC lesions once or twice per day.20,26 In a prospective trial in which 35 children with MC lesions received twice-daily treatments with 10% KOH aqueous solution, complete lesion resolution was observed in 32 of the patients. Applications were discontinued in 3 patients due to severe stinging and secondary infection. The efficacy of KOH has been compared with that of other MC treatments. No significant differences were reported in a trial comparing the efficacy of cryotherapy with that of 10% KOH in solution for the treatment of MC. However, the higher cost and secondary local effects of cryotherapy would tend to favor the use of KOH. Another study found that 10% KOH and 5% imiquimod cream were equally effective, but that KOH had a faster onset of action. Finally, a third study compared 10% KOH administered once per day with salicylic acid and lactic acid in combination, finding they were equally effective in the treatment of MC. Because 10% KOH treatment is noninvasive, efficacious, and can be applied at home, many authors consider it to be the first line of therapy.
MC: Molluscum contagiosum is defined as a contagious viral infection that can manifest on the skin, commonly found on the face in children and on the inner thighs, abdomen, and in adults and athletes.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#2728 Yesterday 00:06:35
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,617
Re: Miscellany
2527) Ammonium Phosphate
Gist
Ammonium phosphate is a group of water-soluble inorganic salts, commonly (triammonium phosphate), formed by reacting ammonia with phosphoric acid. It is a white, crystalline solid with an ammonia odor, primarily used as a high-nutrient, readily absorbed fertilizer (N-P-K), a fire-extinguishing agent, and in food processing.
Ammonium phosphate, including Monoammonium Phosphate (MAP) and Diammonium Phosphate (DAP), is primarily used as a high-nutrient, water-soluble fertilizer, providing nitrogen and phosphorus for agricultural, forestry, and lawn applications. Key uses also include fire suppression in ABC dry chemical extinguishers, food-grade additives (leavening/yeast nutrition), and industrial applications like fire retardants for wood/paper.
Summary
Ammonium phosphate is the inorganic compound with the formula (NH4)3PO4. It is the ammonium salt of orthophosphoric acid. A related double salt, (NH4)3PO4.(NH4)2HPO4 is also recognized but is impractical to use. Both triammonium salts evolve ammonia. In contrast to the unstable nature of the triammonium salts, the diammonium phosphate (NH4)2HPO4 and monoammonium salt (NH4)H2PO4 are stable materials that are commonly used as fertilizers to provide plants with fixed nitrogen and phosphorus.
Details
Ammonium phosphate is an unstable compound made of ammonium and phosphate salt with the chemical formula (NH4)3PO4.
Ammonium phosphate is manufactured by mixing together ammonium phosphate and urea in a molten condition. Considerable heat is generated which transform the ammonium phosphate to the molten state. It includes a group of nitrogen phosphorus materials: mono ammonium phosphates and diammonium phosphates, mixtures of the two or combinations with ammonium nitrate or ammonium sulfate.
Uses of Ammonium phosphate – (NH4)3PO4
* Ammonium phosphate is a broad generic name for a variety of fertilizer materials containing both nitrogen and phosphate.
* Ammonium phosphates are becoming increasingly important as a source of available P2O5. P2O5 is somewhat higher in ammonium phosphates than in triple superphosphate.
* Mainly used as a solid fertilizer but can also be utilized in solution.
* Used as components of intumescent paints and mastics where they function as an acid catalyst.
* Used in paints in which pentaerythritol is the carbonific component and melamine is the specific compound.
Frequently Asked Questions – FAQs
Q1: What is ammonium phosphate used for?
A1: Ammonium phosphate is a high source of elemental nitrogen used as an ingredient in certain fertilizers. This is also used in thermoplastic formulations as a flame retardant.
Q2: What is ammonium phosphate fertilizer?
AmmA2: onium phosphate adds nitrogen and phosphates to the lawns which lack the nutrients. Ammonium phosphate is a fast-release fertilizer that can be used for new grass planting, cleaning, monitoring, or lawn renovation.
Q3: What’s the difference between ammonia and ammonium?
A3: Ammonia contains one nitrogen and three hydrogens opposed to one nitrogen and four hydrogens formed by ammonium. Ammonia is a low, unionized foundation. In the other side, it is ionized to ammonium. Some major distinctions between the two is that Ammonia gives off a heavy odour while Ammonium does not smell at all.
Q4: Is ammonium phosphate an acid?
A4: Ammonium phosphate is an orthophosphoric acid ammonium salt. Formula (NH4)3PO4 is a highly unstable compound. It is elusive, and of little economic interest due to its uncertainty.
Q5: Why is ammonium phosphate soluble in water?
A5: It is soluble in water, and ammonia loses and the acid phosphate (NH4)(H2PO4) is formed as the aqueous solution on the boil. Ammonium phosphate is a high source of elemental nitrogen used as an ingredient in certain fertilizers. This is also used in thermoplastic formulations as a flame retardant.
Additional Information:
Key Properties and Everyday Uses of Ammonium Phosphate
Ammonium phosphate is a salt that is made up of ammonia and phosphorus, and its chemical formula is (NH4)3PO4. However, this is a very unstable salt and due to how unstable it is, it is not exactly a salt worth a lot of commercial value. It can be formed by combining phosphoric acid along with ammonia, or by adding a lot more ammonia with acid phosphate.
For it to be used commercially, it is mostly obtained from crystalline powders.
Major Uses of Ammonium Phosphate:
* Agriculture & Fertilizer: High-concentration, soluble fertilizers (MAP/DAP) provide quick nutrients to plants and are used in foliar feeding and irrigation.
* Fire Retardant & Extinguisher: Used as an ingredient in dry chemical powder extinguishers and in flame-retardant coatings for materials like paper and wood.
* Food Industry: Functions as a leavening agent in baking powders and as a dough conditioner, as well as a yeast nutrient for wine production.
* Industrial Applications: Acts as a fluxing agent in soldering, in the production of intumescent paints, and as a raw material for producing electronics such as piezo-electric crystals.
* Water Treatment: Sometimes used in pH regulation to assist with ammonia removal.
Key Types:
* MAP (Monoammonium Phosphate): Often used for direct soil application, it is highly soluble.
* DAP (Diammonium Phosphate): Widely used for its high nitrogen and phosphorus content.
The Formula of Ammonium Phosphate
The molecular formula for this salt is (NH4)3PO4 and it is also referred to as triammonium phosphate or diazonium hydrogen phosphate.
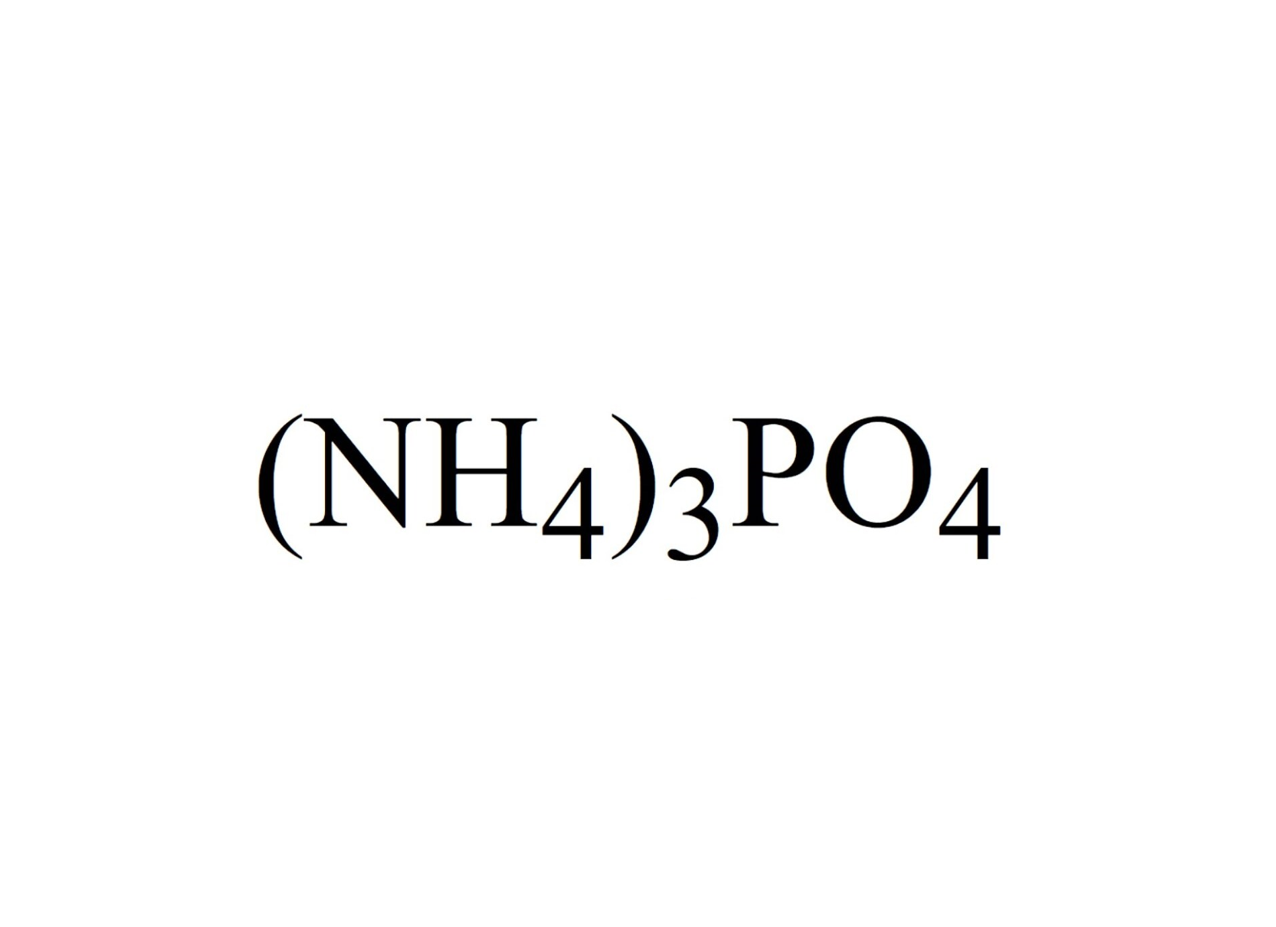
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
#2729 Today 00:46:01
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,617
Re: Miscellany
2528) MASER
Gist
A maser (Microwave Amplification by Stimulated Emission of Radiation) is a device that produces coherent, highly focused electromagnetic waves in the microwave spectrum. Invented in the 1950s, it works by stimulating atoms to emit energy, often used for low-noise amplification in radio telescopes, atomic clocks, and satellite communications.
Masers (Microwave Amplification by Stimulated Emission of Radiation) produce coherent, low-noise microwave signals used for precise timekeeping in atomic clocks, deep-space communication, and high-sensitivity radio astronomy. They are essential for tracking spacecraft, studying interstellar molecular clouds, and providing stable frequency standards for radar.
Summary
A maser is a device that produces coherent electromagnetic waves (microwaves), through amplification by stimulated emission. The term is an acronym for microwave amplification by stimulated emission of radiation. Nikolay Basov, Alexander Prokhorov and Joseph Weber introduced the concept of the maser in 1952, and Charles H. Townes, James P. Gordon, and Herbert J. Zeiger built the first maser at Columbia University in 1953. Townes, Basov and Prokhorov won the 1964 Nobel Prize in Physics for theoretical work leading to the maser. Masers are used as timekeeping devices in atomic clocks, and as extremely low-noise microwave amplifiers in radio telescopes and deep-space spacecraft communication ground-stations.
Modern masers can be designed to generate electromagnetic waves at microwave frequencies and radio and infrared frequencies. For this reason, Townes suggested replacing "microwave" with "molecular" as the first word in the acronym "maser".
The laser works by the same principle as the maser, but produces higher-frequency coherent radiation at visible wavelengths. The maser was the precursor to the laser, inspiring theoretical work by Townes and Arthur Leonard Schawlow that led to the invention of the laser in 1960 by Theodore Maiman. When the coherent optical oscillator was first imagined in 1957, it was originally called the "optical maser". This was ultimately changed to laser, for "light amplification by stimulated emission of radiation". Gordon Gould is credited with creating this acronym in 1957.
Details
A maser is a device that produces and amplifies electromagnetic radiation in the microwave range of the spectrum. The first maser was built by the American physicist Charles H. Townes. Its name is an acronym for “microwave amplification by stimulated emission of radiation.” The wavelength produced by a maser is so constant and reproducible that it can be used to control a clock that will gain or lose no more than a second over hundreds of years. Masers have been used to amplify faint signals returned from radar and communications satellites, and have made it possible to measure faint radio waves emitted by Venus, giving an indication of the planet’s temperature. The maser was the principal precursor of the laser.
A maser oscillator requires a source of excited atoms or molecules and a resonator to store their radiation. The excitation must force more atoms or molecules into the upper energy level than in the lower, in order for amplification by stimulated emission to predominate over absorption. For wavelengths of a few millimetres or longer, the resonator can be a metal box whose dimensions are chosen so that only one of its modes of oscillation coincides with the frequency emitted by the atoms; that is, the box is resonant at the particular frequency, much as a kettle drum is resonant at some particular audio frequency. The losses of such a resonator can be made quite small, so that radiation can be stored long enough to stimulate emission from successive atoms as they are excited. Thus, all the atoms are forced to emit in such a way as to augment this stored wave. Output is obtained by allowing some radiation to escape through a small hole in the resonator.
The first maser used a beam of ammonia molecules that passed along the axis of a cylindrical cage of metal rods, with alternate rods having positive and negative electric charge. The nonuniform electric field from the rods sorted out the excited from the unexcited molecules, focusing the excited molecules through a small hole into the resonator. The output was less than one microwatt (10-6 watt) of power, but the wavelength, being determined primarily by the ammonia molecules, was so constant and reproducible that it could be used to control a clock that would gain or lose no more than a second in several hundred years. This maser can also be used as a microwave amplifier. Maser amplifiers have the advantage that they are much quieter than those that use vacuum tubes or transistors; that is, they add very little noise to the signal being amplified. Very weak signals can thus be utilized. The ammonia maser amplifies only a very narrow band of frequencies and is not tunable, however, so that it has largely been superseded by other kinds, such as solid-state ruby masers.
Solid-state and traveling-wave masers
Amplification of radio waves over a wide band of frequencies can be obtained in several kinds of solid-state masers, most commonly crystals such as ruby at low temperatures. Suitable materials contain ions (atoms with an electrical charge) whose energy levels can be shifted by a magnetic field so as to tune the substance to amplify the desired frequency. If the ions have three or more energy levels suitably spaced, they can be raised to one of the higher levels by absorbing radio waves of the proper frequency.
The amplifying crystal may be operated in a resonator that, as in the ammonia maser, stores the wave and so gives it more time to interact with the amplifying medium. A large amplifying bandwidth and easier tunability are obtained with traveling-wave masers. In these, a rod of a suitable crystal, such as ruby, is positioned inside a wave-guide structure that is designed to cause the wave to travel relatively slowly through the crystal.
Solid masers have been used to amplify the faint signals returned from such distant targets as satellites in radar and communications. Their sensitivity is especially important for such applications because signals coming from space are usually very weak. Moreover, there is little interfering background noise when a directional antenna is pointed at the sky, and the highest sensitivity can be used. In radio astronomy, masers made possible the measurement of the faint radio waves emitted by the planet Venus, giving the first indication of its temperature.
Gas masers
Generation of radio waves by stimulated emission of radiation has been achieved in several gases in addition to ammonia. Hydrogen cyanide molecules have been used to produce a wavelength of 3.34 mm. Like the ammonia maser, this maser uses electric fields to select the excited molecules.
One of the best fundamental standards of frequency or time is the atomic hydrogen maser introduced by American scientists N.F. Ramsey, H.M. Goldenberg, and D. Kleppner in 1960. Its output is a radio wave whose frequency of 1,420,405,751.786 hertz (cycles per second) is reproducible with an accuracy of one part in 30 × 1012. A clock controlled by such a maser would not get out of step more than one second in 100,000 years.
In the hydrogen maser, hydrogen atoms are produced in a discharge and, like the molecules of the ammonia maser, are formed into a beam from which those in excited states are selected and admitted to a resonator. To improve the accuracy, the resonance of each atom is examined over a relatively long time. This is done by using a very large resonator containing a storage bulb. The walls of the bulb are coated so that the atoms can bounce repeatedly against the walls with little disturbance of their frequency.
Another maser standard of frequency or time uses vapour of the element rubidium at a low pressure, contained in a transparent cell. When the rubidium is illuminated by suitably filtered light from a rubidium lamp, the atoms are excited to emit a frequency of 6.835 gigahertz (6.835 × 109 hertz). As the cell is enclosed in a cavity resonator with openings for the pumping light, emission of radio waves from these excited atoms is stimulated.
Additional Information
MASER stands for Microwave Amplification by Stimulation Emission of Radiation. A LASER is a MASER that works with higher frequency photons in the ultraviolet or visible light spectrum (photons are bundles of electromagnetic energy commonly thought of as "rays of light" which travel in oscillating waves of various wavelengths) .
The first papers about the MASER were published in 1954 as a result of investigations carried out simultaneously and independently by Charles Townes and co-workers at Columbia University in New York and by Dr. Basov and Dr. Prochorov at the Lebedev Institute in Moscow. All three of these gentlemen received the Nobel Prize in 1964 for their contributions to science.
[The following was paraphrased in part from Halliday & Resnick's "Fundamentals of Physics", second edition.]
The fundamental physical principle motivating the MASER is the concept of stimulated emission, first introduced by Einstein in 1917. Before defining it we look at two related but more familiar phenomena involving the interplay between matter and radiation, absorption and spontaneous emission.
* Absorption. According to quantum mechanics, absorption of photons by atoms occurs only if the wavelength of the photon is just the right size (say, of wavelength l). If it is, the atom will "absorb" it (the photon vanishes) and go to a higher energy state. In physics, this process is called "absorption."
* Spontaneous Emission. Atoms don't like to stay in high energy states (this is dictated by the laws of thermodynamics), so after absorbing a photon and going to a higher energy state, they will move of their own accord to a lower energy state, emitting a photon in the process. This is called "spontaneous emission" because no outside influence triggers the emission. Normally the average lifetime for spontaneous emissions by excited atoms is around 10-8 seconds (that is, the atom or molecule will usually take around 10-8 seconds before emitting the photon). Occasionally, however, there are states for which the lifetime is much longer, perhaps around 10-3 seconds. These states are called metastable. Metastable emission levels are essential for a working MASER and will be discussed further in a moment.
Now that we've discussed absorption and spontaneous emission, we can get to stimulated emission (a MASER beam is made up entirely of stimulated emission).
* Stimulated Emission. With stimulated emission, a photon of the absorption wavelength, l , is fired at an atom already in its high energy state from prior absorption. The atom absorbs this photon, and then quickly emits two photons to get back to its lower energy state. Thanks to quantum mechanics, both of these newly emitted photons are of wavelength l! The following figure displays this concept in detail:
* MASER. In each frame, a molecule in the upper level of the MASER transition (that is, in the high energy, excited state) is indicated by a large red circle, while one in the lower level (low energy state) is indicated by a small blue circle. (a) All of the molecules are in the upper state and a photon of wavelength l (shown in green) is incident from the left. (b) The photon l stimulates emission from the first molecule, so there are now two photons of wavelength l, in phase. (c) These photons stimulate emission from the next two molecules, resulting in four photons of wavelength l. (d) The process continues with another doubling of the number of photons.
Basically, a man-made MASER is a device that sets up a series of atoms or molecules and excites them to generate the chain reaction, or amplification, of photons. Metastable emission states make MASERs and LASERs possible. To get the proper wavelengths to generate the chain reaction, first electricity or another energy source is "pumped" into a chamber filled with particular atoms or molecules. Then this "pumping" radiation causes the transition of atoms from the ground state to a high energy excited state higher than that referred to in the above paragraphs. From this short-lived state the atoms come down through non-radiative transition to the long-lived metastable state. Once in the metastable state many atoms can be accumulated in one place and in the same state. The LASER or MASER beam, stimulated emission, arises when all these accumulated atoms simultaneously make a transition to the ground state, releasing their energy of wavelength l, creating a beam of microwave radiation (or visible light in the case of a LASER) which can be sent on to other atoms to cause the chain reaction described in the above figure. Since all the resulting photons are the same wavelength, MASER beams are extremely focussed and coherent. MASERs and their shorter-wavelength counterparts (LASERs), have many practical applications, especially in science and medicine.
Naturally occurring MASERs have been discovered in interstellar space. For more information about MASERs in space, check out this site for a discussion of astrophysical MASERs.

It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline