Math Is Fun Forum
You are not logged in.
- Topics: Active | Unanswered
Pages: 1
#1 Today 00:17:40
- Jai Ganesh
- Administrator

- Registered: 2005-06-28
- Posts: 53,737
Calcination
Calcination
Gist
Calcination is a thermal treatment process that heats raw materials—typically minerals or ores—to high temperatures (below their melting point) in a limited supply of oxygen or absence of air. It decomposes carbonates, removes volatile organic matter, and drives off moisture, commonly used for producing lime, alumina, and cement.
What causes calcination?
When limestone (CaCO3) is heated in a kiln, it undergoes a chemical reaction called calcination. This process produces calcium oxide (CaO) and releases CO2 as a byproduct. Calcination accounts for almost two-thirds of the total CO2 emissions from cement production.
Summary
Calcination is the heating of solids to a high temperature for the purpose of removing volatile substances, oxidizing a portion of mass, or rendering them friable. Calcination, therefore, is sometimes considered a process of purification.
A typical example is the manufacture of lime from limestone. In this process the limestone is brought to a temperature high enough to expel the carbon dioxide, producing the lime in a highly friable or easily powdered condition. Calcination in special cases may be carried on in furnaces designed to exclude air, for which an inert gas may be substituted.
What is another name for calcination?
Although calcination in the strict sense only means decarbonisation or dehydration, today it is used to refer to a wider set of thermal processes that involve high-temperature treatments. The goal may be to heat solid materials to dehydrate them, decompose them or transform them chemically.
Details
Calcination is thermal treatment of a solid chemical compound (e.g. mixed carbonate ores) whereby the compound is raised to high temperature without melting with a restricted supply of oxygen (i.e. gaseous O2 fraction of air), generally for the purpose of removing impurities or volatile substances and/or to induce thermal decomposition.
The root of the word calcination refers to its most prominent use, which is to remove carbon and oxygen from limestone (calcium carbonate) through applying heat to yield calcium oxide (quicklime). This calcination reaction (which is endothermic) is CaCO3(s) → CaO(s) + CO2(g).
Calcium oxide is a crucial ingredient in modern cement, and is also used as a chemical flux in smelting. Industrial calcination generally emits carbon dioxide (CO2).
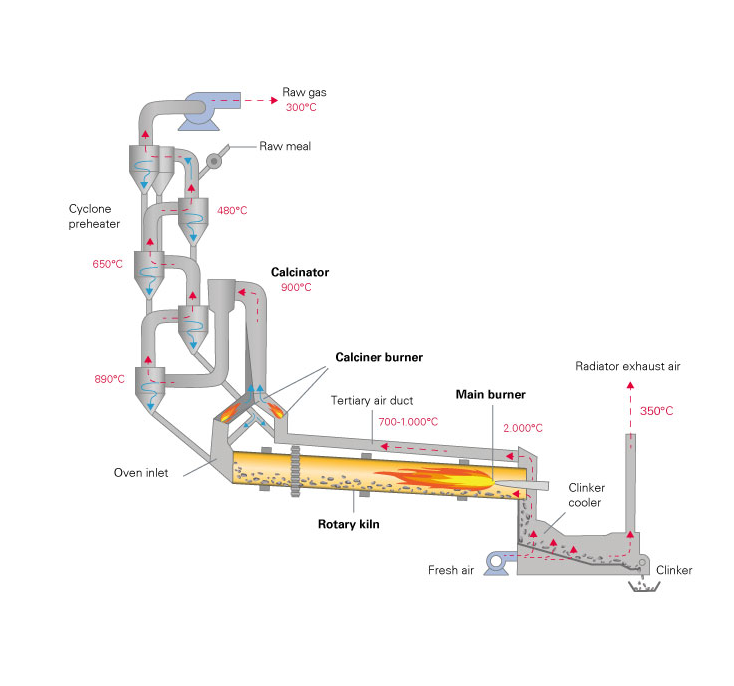
A calciner is a steel cylinder that rotates inside a heated furnace and performs indirect high-temperature processing (550–1150 °C, or 1000–2100 °F) within a controlled atmosphere.
Calcining is simply firing a ceramic material to create a powder of new physical properties. Often it is done to kill the plasticity or burn away the hydrates, carbonates, sulfates of a clay or refractory material.
The calcining process is most commonly used to remove some or all unwanted volatiles from a material (e.g. H2O, CO2, SO2 - thus eliminating the LOI) and/or to convert a material into a more stable, durable or harder state. However, in ceramics, it is also useful to destroy the plasticity of a clay for use in glazes (non-plastic clays reduce drying shrinkage). Varying temperatures are employed to calcine materials, depending on the decomposition temperature of the volatiles being burned out or the degree of sintering (and thus physical characteristics change) needed.
The cement industry is by far the largest consumer of calcined clay. And the largest calcined powder producers. They heat-treat the kaolin in rotary kilns at 1450C (mixed with limestone and iron ore to form clinkers ground with gypsum to get the final product).
To produce molochite, lump kaolins are calcined at high temperatures (but lower than that of the cement process), they are then ground and sized. Powdered calcined kaolin is burned at lower temperatures.
Potters typically calcine clays to enable using them in higher percentages in glazes, slips and engobes (the electrolytics of clay particles are destroyed by this process resulting in less shrinkage while drying). Typical calcine temperatures are not necessary to accomplish this, only about 1000F is needed (cone 022 or red heat). At this temperature, the process is commonly called "roasting". The loose powder can be fired in bisque vessels made of pottery clay (any clay can easily withstand this temperature. For large or heavy-walled roasting vessels, fire slower (e.g. 200F per hour). For small amounts, 500F/hr should be fine. Hold at temperature for the time necessary for the heat to penetrate (start with 30 minutes). If any black powder remains in the center extend the soak time next firing.
The calcining and roasting processes produce a material having no LOI (Loss on Ignition), if it is being substituted into a glaze this needs to be considered. For example, if a kaolin loses 12% weight on firing, then 12% less of the calcine would be needed in the glaze recipe.
Calcining can actually produce a less stable form of certain materials, they gradually want to revert to the former carbonated or hydrated state. For a good example of this, mix calcium carbonate with kaolin and make a bar and fire it. Out of the kiln, it will appear to be a hard ceramic. But after several days it will absorb CO2 from the air and completely fracture into a powder. Pour water on it and it will immediately fracture and generate considerable heat as it disintegrates.
Calcined clays are not normally used as ingredients for traditional clay bodies (because of cost and plasticity loss). But for refractories and high-tech product manufacture the use of calcined materials is common.
Additional Information
The Calcination is a thermal treatment process in presence of air or oxygen; applied to ores and other solid materials to bring about a thermal decomposition, phase transition, or removal of a volatile fraction. The calcination process usually takes place at temperatures below the melting point of the product materials. Materials processed are rare earths, precious metals, refractory metals, pigments and phosphor. Vesuvius provides trays and containers for calcination processes performed in kilns at temperatures below 1200°C.
* Higher cycling rate Unique fused silica material enables repeated strong thermal cycling through excellent resistance to thermal shock.
* Strong resistance to breakage in operations Whether your process is robotic or manual to load and to pour your powders, the high mechanical resistance of the part avoids breakage and makes its lifetime longer, compared to other materials.
* No powder contamination The selection of dedicated raw material and the high resistance of fused silica material to chemical corrosion matches the strong purity requirements in some applications.
* No process interruption Precise bottom flatness ensures tray stability on all conveying systems.
It appears to me that if one wants to make progress in mathematics, one should study the masters and not the pupils. - Niels Henrik Abel.
Nothing is better than reading and gaining more and more knowledge - Stephen William Hawking.
Offline
Pages: 1